Comment:
The summary of this review is striking for the apparent disconnect it highlights regarding Desiccated Thyroid Extract (DTE). The authors accurately report that official guidelines universally reject DTE based on theoretical concerns—namely, the lack of FDA oversight and the transient, non-physiological T3 spikes. Yet the clinical data they reviewed tells an entirely different story: clinical studies found “no reported ADRs,” “no cardiovascular harm,” and similar normalization of TSH and symptoms when DTE was dosed appropriately.
This stark contrast perfectly underlines how institutional bias can triumph over available evidence. The potential for a theoretical cardiac effect from a T3 spike, or a concern about a drug’s regulatory status, is apparently given more weight than years of clinical trial data and long-term observational studies showing no actual harm. For patients who struggle on standard levothyroxine (LT4) therapy, the collective refusal of guidelines to acknowledge the clinical reality—that DTE, when managed correctly, appears safe and effective—is a failure to prioritize patient-centered care.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
🧐Clinical Bottom Line
This review article affirms that levothyroxine (LT4) monotherapy is the universal, first-line treatment for newly diagnosed hypothyroidism. The authors acknowledge, however, that a subset of patients remains symptomatic on LT4, possibly because LT4 monotherapy does not always restore T3 levels to the same state as a healthy thyroid.
For these patients with persistent symptoms despite a normal TSH, the authors conclude that a trial of combination LT4 + liothyronine (LT3) therapy is a reasonable approach. They cite data from nearly 1,000 patients in clinical trials and a long-term observational study, which found this combination (when dosed to a normal TSH) did not increase the risk of cardiovascular events, atrial fibrillation, or fractures compared to LT4 alone.
Summary of Findings
-
LT4 Monotherapy: This is the standard of care recommended by all clinical guidelines. However, the authors note that in LT4-treated patients, T3 levels are often lower and T4 levels are higher than in healthy controls, which may explain persistent symptoms in some individuals.
-
LT4 + LT3 Combination Therapy: For patients who have “unambiguously not benefited from LT4,” a trial of combination therapy is supported by several thyroid associations (BTA, ETA, ATA). A recommended starting point is to reduce the LT4 dose by 25 mcg/day and add 2.5–7.5 mcg of LT3 once or twice daily.
-
DTE (Desiccated Thyroid Extract): This is a form of combination therapy, typically from porcine thyroids, with an LT4:LT3 ratio of approximately 4:1.
-
LT3 Monotherapy: The authors conclude there is no practical or conceptual reason to use LT3 alone for long-term hypothyroidism treatment. The doses required to normalize TSH (30-45 mcg/day) cause significant serum T3 spikes above the normal range, and the short half-life makes it impractical.
Analysis of DTE: Guideline Concerns vs. Presented Data
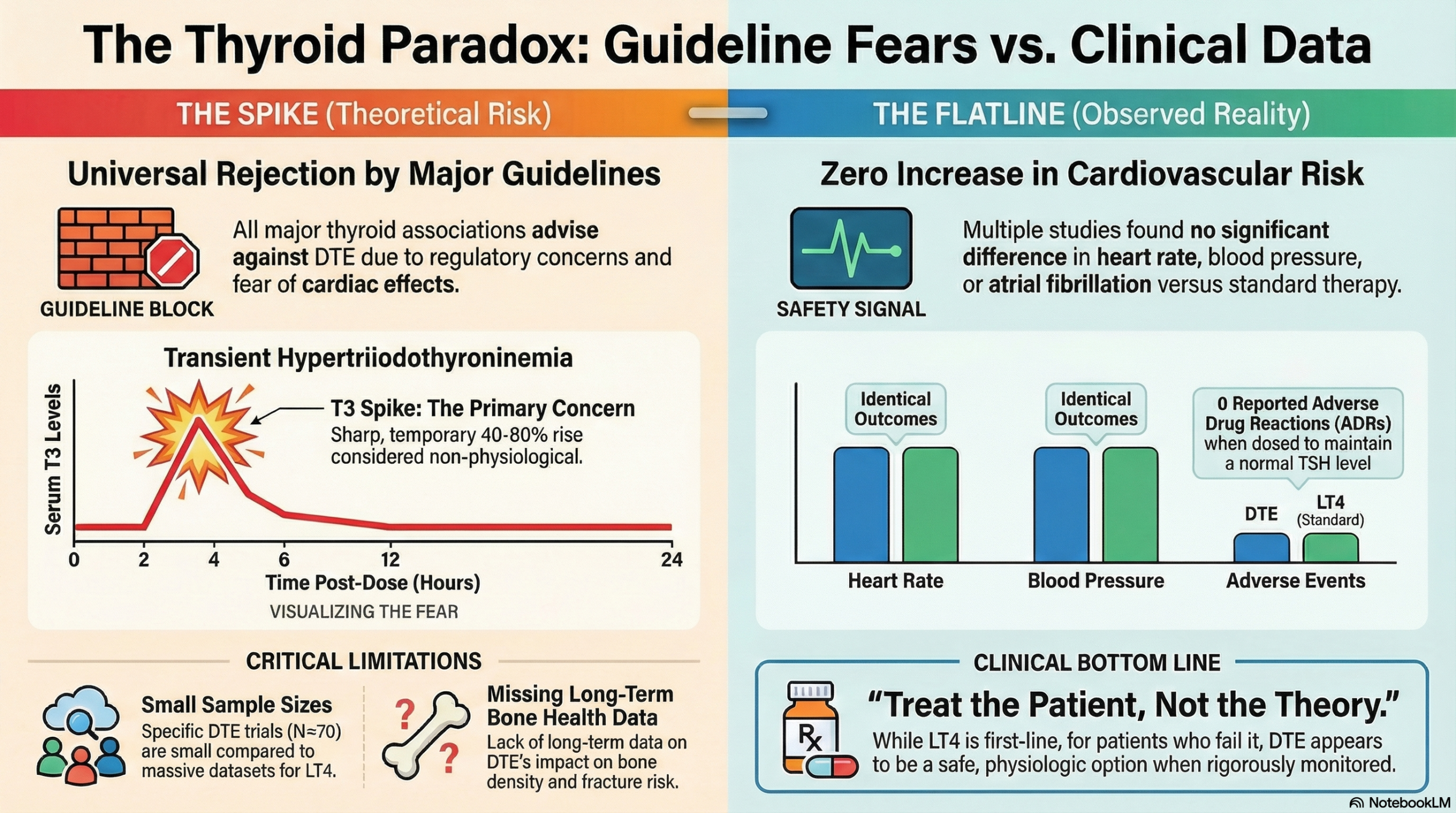
The authors report that DTE is universally not recommended by clinical guidelines, while simultaneously presenting a summary of clinical studies in which no adverse effects were found.
1. Stated Concerns and Warnings (The “Warnings”)
-
Lack of FDA Oversight: DTE is a non-FDA-approved drug. Its consistency and T4/T3 content are monitored only by the manufacturers, not by an independent body.
-
Pharmacokinetic Spikes: DTE causes a daily, transient spike in serum T3 levels about 2–3 hours after ingestion. At higher doses, this T3 peak can rise above the normal reference range.
-
Guideline Non-Recommendation: The authors clearly state that the AACE, ATA, BTA, ETA, and NICE guidelines do not recommend the use of DTE. The American Geriatrics Society also lists it as potentially inappropriate for older adults due to “concerns about cardiac effects”.
-
Missing Long-Term Data: The authors note that long-term assessment of bone density is not available for DTE-treated patients.
2. Presented Clinical Trial Data (The “Data”)
In contrast to these warnings, the clinical studies reviewed by the authors that actually used DTE and monitored patients failed to report significant adverse events.
-
No Reported ADRs: In their own summary of the DTE literature, the authors state that DTE “restores clinical and biochemical euthyroidism with no reports of ADRs, as long as fasting serum TSH levels are used to adjust the replacement dose”.
-
No Cardiovascular Harm Found: The authors explicitly conclude that “the clinical euthyroidism and absence of cardiovascular ADRs in all reported patients indicate that… a daily transient elevation in serum T3 levels was not associated with cardiovascular ADRs“.
-
Specific Study Findings:
-
A comparative study found DTE normalized TSH and symptoms similarly to LT4, with no ADRs.
-
A A 70-patient randomized, double-blind, crossover study comparing DTE to LT4 found no differences in heart rate, blood pressure, or symptoms, and no ADRs were reported.
-
A 6-year observational study found that DTE use did not result in additional risk of atrial fibrillation, cardiovascular disease, or mortality.
-
A Framingham Heart Study analysis of older adults (mean age 68.6), including 122 on DTE for an average of 19 years, made no mention of thyrotoxic symptoms.
-
In summary, the authors accurately report a major disconnect: the official clinical guidelines and theoretical concerns (lack of FDA oversight, T3 spikes) stand in sharp contrast to the available, albeit limited, clinical data, which fails to show evidence of harm when DTE is dosed appropriately to a normal TSH.
Bibliographic Data
-
Title: Liothyronine and Desiccated Thyroid Extract in the Treatment of Hypothyroidism
-
Authors: Thaer Idrees, Scott Palmer, Rui M.B. Maciel, and Antonio C. Bianco
-
Journal: Thyroid
-
Year: 2020
Original Article:
Full text: PubMed Central
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
