Comment:
This study is a classic example of one that is both exciting and deeply frustrating.
The exciting part: A simple, cheap intervention (melatonin) was associated with a 70% relative reduction in death or heart failure readmission after a heart attack, supported by a significant, positive change in the MMP-9 biomarker.
The frustrating part: The study is a methodological mess. It’s a small pilot trial with significant baseline imbalances. Most critically, the authors describe it as “open-label” while also saying they used a “placebo”—a major contradiction. They also fail to report any safety data or break down the components of the primary endpoint.
My take: These results are intriguing and certainly justify a larger, better-designed trial. But for now, this is purely hypothesis-generating and should not change clinical practice.
Summary:
Clinical Bottom Line
This small, single-center pilot study suggests that a single intravenous infusion of melatonin given just before primary percutaneous coronary intervention (PCI) for acute myocardial infarction (AMI) is associated with a reduction in the composite rate of death or heart failure readmission at 2 years. This clinical finding was supported by a corresponding significant reduction in post-procedural levels of MMP-9, a biomarker linked to cardiac remodeling.
However, these findings should be considered hypothesis-generating and not practice-changing at this time. The study is limited by its small size (n=94), significant baseline imbalances between groups despite randomization, and a critical lack of clarity regarding blinding. Furthermore, the unadjusted result for the primary endpoint was not statistically significant (HR 0.3, 95% CI 0.08–1.08), though a post-hoc adjusted analysis did reach statistical significance.
Results in Context
📈 Primary Outcome
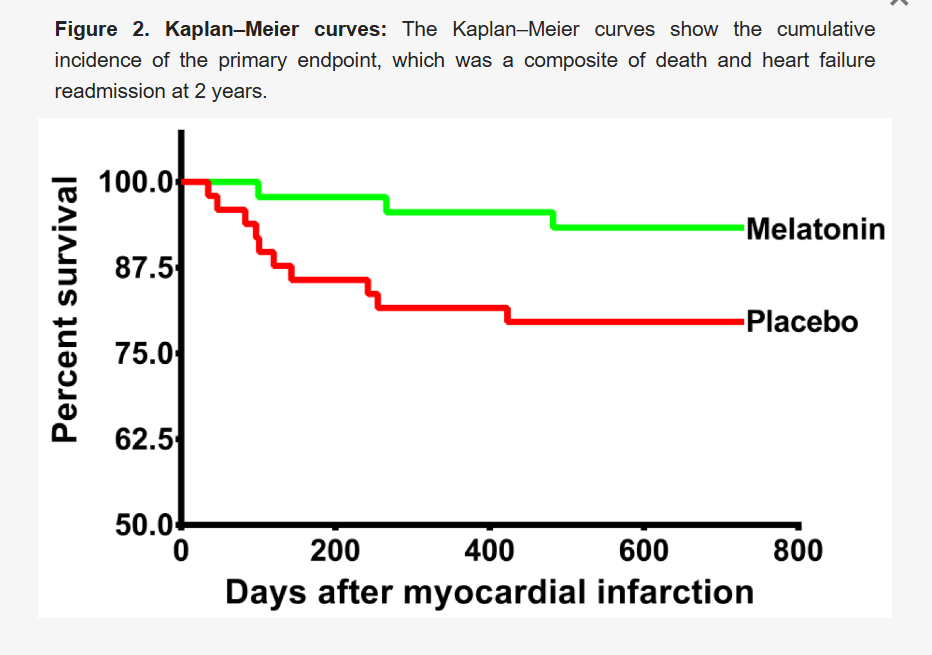
The primary endpoint was a composite of all-cause mortality or readmission due to heart failure at a 2-year follow-up.
-
Events: This composite event occurred in 3 of 45 patients (6.7%) in the melatonin group compared to 10 of 49 patients (20.4%) in the placebo group.
-
Hazard Ratio (HR): The unadjusted HR was 0.3 (95% CI 0.08–1.08; p = 0.06). A hazard ratio of 0.3 suggests a 70% reduction in the hazard of the event occurring in the melatonin group. However, this result was not statistically significant, and the 95% confidence interval (CI) just crossed 1.0, indicating the possibility of no benefit or even slight harm.
-
Adjusted Analysis: After statistically adjusting for baseline differences (hemoglobin and number of diseased vessels), the adjusted HR was 0.27 (95% CI 0.08–0.97; p = 0.046), which is a statistically significant finding.
-
Restricted Mean Survival Time (RMST): The analysis also showed a statistically significant difference in RMST of 87.5 days (p = 0.02). This means that, on average, patients in the melatonin group lived 87.5 days longer without the primary event over the 2-year period than those in the placebo group.
🔬 Key Secondary & Specialized Outcomes
The secondary endpoint was the level of Matrix Metalloproteinase-9 (MMP-9), an enzyme involved in tissue remodeling after a cardiac event, measured just after the PCI.
-
The melatonin group had significantly lower median MMP-9 levels compared to the placebo group: 142 ng/mL vs. 248 ng/mL (p < 0.001).
-
This finding supports the study’s biological hypothesis that melatonin may exert a cardioprotective effect by attenuating the MMP-9 inflammatory response to ischemia-reperfusion injury.
⚠️ Harms and Adverse Events
The article fails to report any information on adverse events or harms related to either the melatonin or placebo infusion. This is a significant omission, as it provides no data on the safety or tolerability of the intervention.
Assertive Critical Appraisal
⚖️ Risk of Bias (RoB 2 Framework)
Overall judgment: Some concerns.
-
Randomization: The study was randomized, but the process was not described. Despite randomization, significant baseline imbalances were present between the two small groups. Notably, the placebo group had lower weight, lower hemoglobin levels, and a higher prevalence of 2- or 3-vessel coronary disease. These differences could confound the results and are the likely reason the authors performed a post-hoc adjusted analysis.
-
Blinding: The methodology contains a major contradiction. It describes the study as “open-label” (unblinded) but also states that patients were randomized to receive either melatonin or “placebo (50 mL physiological saline)”. The use of a placebo strongly implies blinding of patients and, potentially, clinicians. This lack of clarity is a critical methodological flaw. If the trial was indeed open-label, it introduces a high risk of performance and detection bias.
-
Missing Data: The study reports no patients were lost to follow-up for the primary endpoint, which is a major strength.
📦 Composite Endpoints
The primary endpoint was a composite of death and heart failure readmission.
-
Deconstruction: The 13 total events were composed of 3 deaths and 10 readmissions due to heart failure.
-
Critical Flaw: The paper fails to report the breakdown of these components by treatment group. We do not know how many deaths occurred in the melatonin group versus the placebo group. It is possible the entire effect was driven by the less severe endpoint (HF readmission). This lack of transparency is a serious reporting failure.
📋 Reporting Quality Assessment (CONSORT)
The paper’s reporting has significant deficiencies.
-
Participant Flow: A CONSORT flow diagram is included (Figure 1), which is a strength.
-
Missing Methods: The authors do not describe the method of randomization (e.g., sequence generation) or, crucially, allocation concealment. This prevents a full assessment of selection bias.
-
Missing Harms: As noted, the complete absence of safety and adverse event reporting is a major violation of CONSORT principles.
Applicability
This study investigates a potentially simple, inexpensive, and widely available intervention (melatonin) for a common and serious condition (AMI). The intervention is feasible. However, this study’s role is strictly as a pilot study. Its small size, methodological contradictions, and baseline imbalances mean the results are not robust enough to support any change in clinical practice. They do, however, provide a strong rationale and the necessary preliminary data to design a larger, multicenter, and more methodologically sound randomized controlled trial.
Research Objective
To assess whether early intravenous melatonin, given immediately before reperfusion, improves clinical outcomes (death or heart failure readmission at 2 years) and reduces MMP-9 levels in patients with ST-segment elevation AMI (STEMI) successfully treated with primary PCI.
Study Design
-
Design: A “prospective, randomized, open-label study” that was also described as placebo-controlled.
-
Allocation: 94 patients were randomized. 45 were allocated to the melatonin group and 49 to the placebo group. Analysis was per the “intention to treat” principle.
-
Intervention: A 60-minute intravenous infusion of either melatonin (12 mg in 50 mL) or placebo (50 mL physiological saline) started immediately before primary PCI.
Setting and Participants
-
Setting: Patients presenting with STEMI undergoing primary PCI.
-
Participants: 94 patients were randomized from 675 assessed for eligibility.
-
Inclusion Criteria: STEMI, with symptom onset within 3 hours of treatment initiation.
-
Exclusion Criteria: Included age <18 or >85, severe renal or hepatic failure, cardiogenic shock, and cardiac arrest.
📚 Bibliographic Data
-
Title: Early Treatment of Acute Myocardial Infarction with Melatonin: Effects on MMP-9 and Adverse Cardiac Events
-
Authors: Domínguez-Rodríguez, A.; Hernández-Vaquero, D.; Abreu-González, P.; et al.
-
Journal: Journal of Clinical Medicine
-
Year: 2022
-
DOI: 10.3390/jcm11071909
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
