Comment:
This is a classic “real-world evidence” abstract that generates a frightening headline—a 90% increased risk of heart failure!—but whose findings are likely the result of a fatal flaw: confounding by indication.
It’s impossible to fully judge a study from a conference abstract alone, as the methodology is never described in sufficient detail so we’ll have to wait for the full publication. However, the core problem is the study compares two fundamentally different groups of people. The “melatonin group” isn’t just people taking melatonin; it’s people with insomnia so severe, chronic, or refractory that it required a long-term prescription. This group is almost guaranteed to be sicker, frailer, and have more unmeasured confounders (like severe sleep apnea or psychiatric comorbidities) than the control group.
The authors’ use of propensity-score matching is a valiant effort, but it can only control for the 40+ variables measured and available in the database. It cannot control for the reason a physician prescribed the drug, which is likely related to unmeasured confounders.
Summary:
Clinical Bottom Line
This large, real-world cohort study shows a strong statistical association between long-term (≥ 1 year) prescribed melatonin and a markedly increased risk of heart failure and mortality. However, this study suffers from a critical and likely fatal flaw: confounding by indication. It is highly probable that the severe, negative outcomes observed are not caused by the melatonin but are instead a feature of the sicker, more complex patients who are selected for long-term prescription melatonin. This observational study cannot prove causation, and its findings should be viewed with extreme skepticism.
Results in Context
Main Results
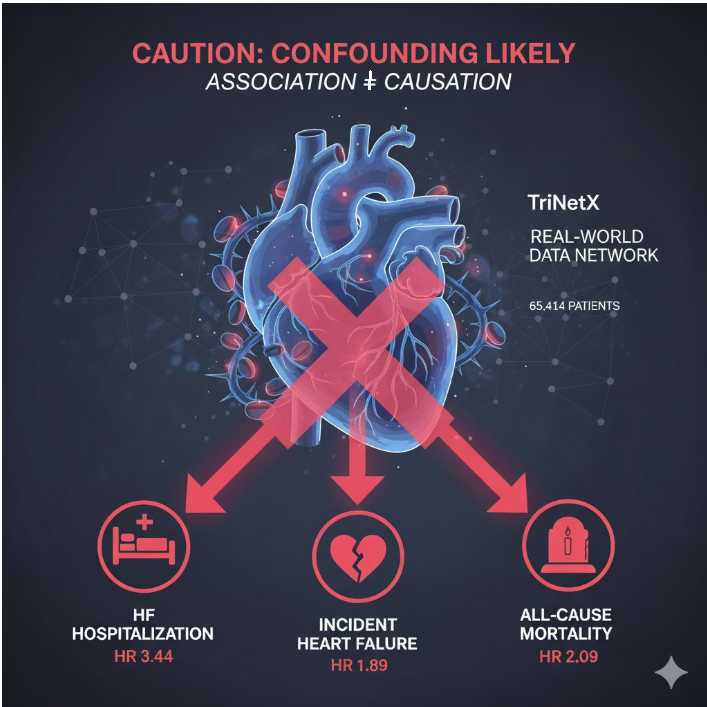
The study compared 65,414 propensity-score matched melatonin users to 65,414 controls over 5 years.
-
Incident Heart Failure: 4.6% in melatonin users vs. 2.7% in controls.
-
Hazard Ratio (HR) = 1.89 (95% CI 1.78–2.00)
-
-
HF Hospitalization: 19.0% in users vs. 6.6% in controls.
-
Hazard Ratio (HR) = 3.44 (95% CI 3.32–3.56)
-
-
All-Cause Mortality: 7.8% in users vs. 4.3% in controls.
-
Hazard Ratio (HR) = 2.09 (95% CI 1.99–2.18)
-
Definitions
-
A Hazard Ratio (HR) indicates the relative hazard of an event. An HR of 1.89 means the melatonin group had an 89% higher hazard (or risk) of developing heart failure at any point during follow-up compared to the control group, assuming all other risk factors are equal—an assumption which is highly questionable here.
Participants: The analysis included 130,828 adults (65,414 matched pairs) with an insomnia diagnosis.
Assertive Critical Appraisal
Limitations & Bias (STROBE Framework)
The primary threat to this study’s validity is unmeasured confounding. While the authors’ use of propensity-score matching on over 40 variables is a sophisticated attempt to address this (and represents high-quality reporting), it cannot overcome the fundamental problem.
-
Propensity-score matching can only control for measured confounders—factors that are known and well-coded in the database (e.g., demographics, 15 comorbidities, labs, vitals).
-
The method cannot control for unmeasured confounders, which are likely the true drivers of the results.
-
**The most critical unmeasured confounder is confounding by indication (or severity). One must ask: Why does one patient with an insomnia diagnosis get a long-term prescription for melatonin, while another (the control) does not? The “exposed” group almost certainly has more severe, chronic, or refractory insomnia. This underlying severity is likely a proxy for poorer overall health, higher frailty, more severe (though unrecorded) sleep apnea, or more significant co-existing psychiatric illness—all of which are powerful, independent risk factors for both heart failure and mortality.
-
The study is not comparing melatonin to no-melatonin in similar patients; it is comparing patients who require prescription melatonin to patients who do not, and then attempting to statistically correct for the differences. The very large hazard ratios (HR > 2.0) are a strong signal that this statistical adjustment likely failed, and the groups remained fundamentally different in their underlying risk.
Reporting Quality Assessment (STROBE)
In accordance with the STROBE statement, the authors have clearly described their efforts to address confounding (propensity-score matching). However, this is a critical flaw, as the abstract does not (and perhaps cannot) address the unmeasured confounders that are the most likely explanation for the findings. The conclusion that melatonin itself is associated with this degree of harm is not supported by this study design.
Reporting Quality Assessment (RECORD) for RWE Studies
The abstract follows the RECORD statement by clearly describing the data source (TriNetX), participant selection (ICD/prescription codes), and outcome definitions (ICD codes). This transparency is good, as it allows us to identify the inherent limitations of this real-world data source—namely, its inability to capture the clinical reason for the prescription or the underlying severity of the patient’s condition.
Applicability
The findings are not applicable to the general population using short-term or over-the-counter (OTC) melatonin, as the study specifically selected for patients with a prescription for long-term use (≥ 1 year). The results are likely relevant only as an illustration of the poor prognosis of the patient population that receives long-term prescribed melatonin, not as an indicator of drug toxicity.
Research Objective
To assess the long-term cardiovascular safety (incident heart failure, HF-related hospitalization, and all-cause mortality) associated with long-term melatonin supplementation in adults with an insomnia diagnosis.
Study Design
This is a retrospective, propensity-score-matched cohort study using the TriNetX multinational real-world database.
Setting and Participants
-
Setting: Multinational electronic health record database.
-
Participants: 65,414 matched pairs of adults (age ≥ 18) with an ICD-10 diagnosis of insomnia.
-
Exclusions: Patients with prior heart failure or those using other prescription hypnotics.
Bibliographic Data
-
-
Title: Effect of Long-term Melatonin Supplementation on Incidence of Heart Failure in Patients with Insomnia
-
Authors: Ekenedilichukwu Nnadi, MD, Maureen Masara, MD, Rita Offor, MD, Selin Unal, MD, Rebhi Rebah, MD, Muhammed Atere, MD, Bisrat Nigussie, MD, and Suzette Graham-Hill, MD
-
Journal: Circulation – Abstracts From the American Heart Association’s 2025 Scientific Sessions and the American Heart Association’s 2025 Resuscitation Science Symposium
-
Year: 2025 (Abstract supplement, Vol. 152)
-
Mandatory Disclaimer: This AI-generated analysis is for informational and research purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of a qualified health provider with any questions you may have regarding a medical condition.
Original Article:
Abstract: here
