Comment:
I don’t usually review abstracts, but I think this is interesting enough that it’s worth posting at the very least to mark the dosing strategy, which finally mimics what we do clinically. The most compelling takeaway from the TARGET-D trial isn’t just the reduction in recurrent Myocardial Infarction (MI), but how they achieved it: through a “treat-to-target” dosing strategy.
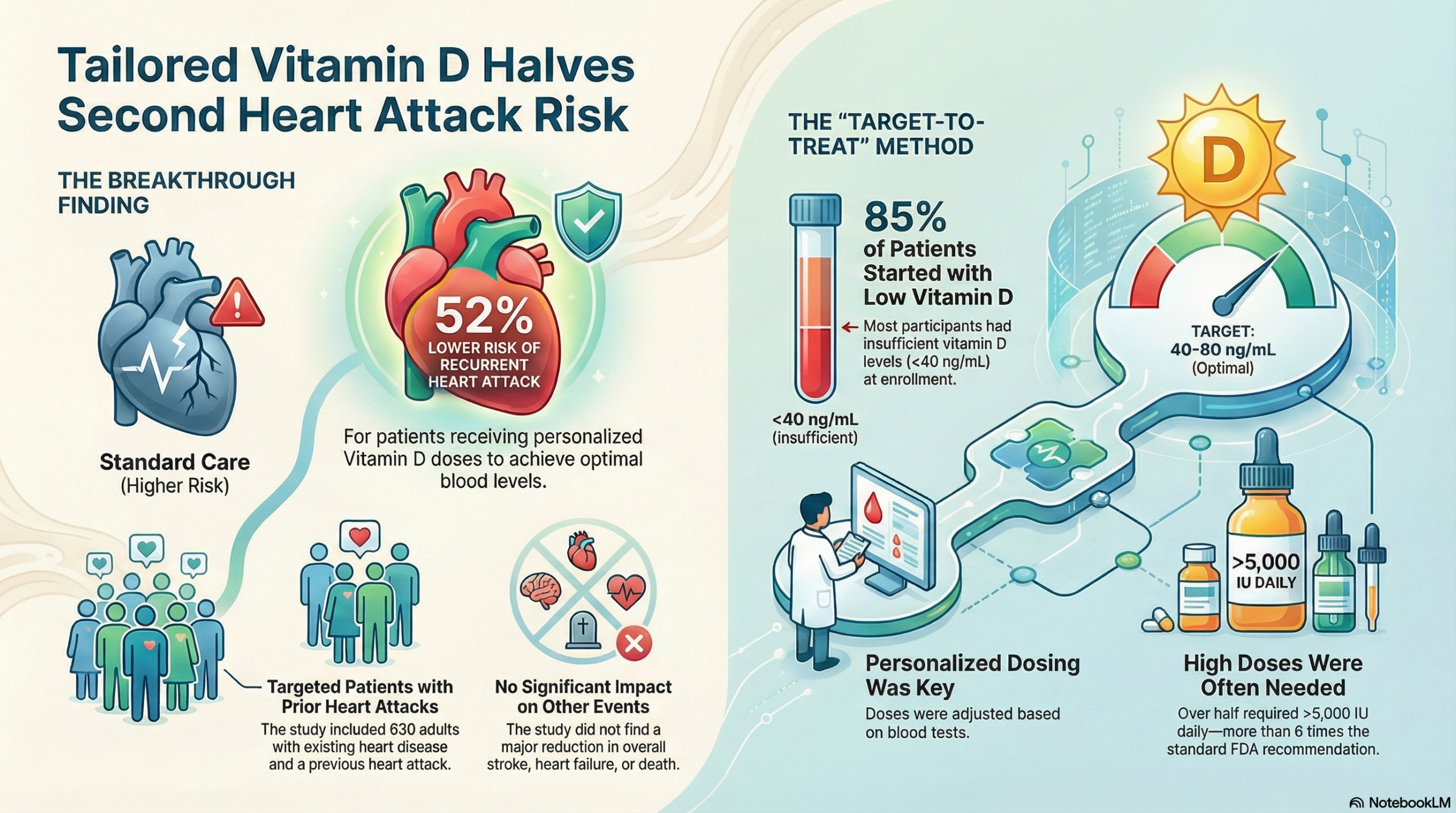
This is huge because it validates what we already know from a clinical perspective and what various researchers have been observing: the one-size-fits-all “blanket” dosing used in previous negative trials (like VITAL and D-Health) is fundamentally flawed.
Here’s why this individualized approach in TARGET-D is so critical and aligns with real-world practice:
-
Massive Dosing Heterogeneity Required: The trial found that 87% of their post-ACS patients were deficient (<40 ng/mL). To get them to the target level, 58.8% required a starting dose of 5,000 IU daily—a dose significantly higher than the standard Recommended Dietary Allowance (RDA) of 600-800 IU and those used in other clinical trials. but in alignment with what clinicians who use vitamin D know to be true. The study and graph I go back to again and again is here.
-
The Need for Titration is Key: The TARGET-D protocol required frequent testing and dose adjustment (titration) until the therapeutic range of 40-80 ng/mL was reached. This directly reflects the rigorous approach often necessary in clinical practice to actually see biomarker correction, a strategy that previous large-scale studies failed to employ.
-
A Plausible Mechanism for the MI Reduction: By successfully correcting the deficiency with an aggressive, individualized dose, the TARGET-D trial may have finally delivered enough Vitamin D3 to activate its non-skeletal, anti-inflammatory and endothelial protective effects in the heart. The significant 52% relative risk reduction in follow-up MI—a hard, objective outcome—provides a strong, hypothesis-generating signal that targeted normalization, not just generalized supplementation, might actually confer a cardiovascular benefit in this high-risk population.
While we still need the full manuscript to fully vet the primary outcome’s non-significance against the strong secondary MI finding, this trial provides essential evidence: If you want a vitamin D trial to test a hypothesis about optimal levels, you must first ensure you actually achieve those levels.
Summary:
Clinical Bottom Line
The TARGET-D trial introduces a novel “treat-to-target” dosing strategy for vitamin D, distinguishing it from previous trials that failed using fixed “blanket” doses. While the study successfully normalized vitamin D levels in post-Acute Coronary Syndrome (ACS) patients, it did not achieve statistical significance for its primary composite outcome (Major Adverse Cardiovascular Events – MACE).
However, the study identified a significant and clinically substantial reduction in recurrent Myocardial Infarction (MI)—a key secondary component—with the treatment arm showing a 52% relative risk reduction compared to standard care. Because this is an isolated positive finding within a negative primary composite from a preliminary conference abstract, clinicians should view this as a strong, hypothesis-generating signal warranting confirmation in a larger trial, rather than immediate grounds for changing practice.
Results in Context
Primary Outcome
The pre-specified primary composite outcome was MACE (death, MI, heart failure hospitalization, and stroke).
-
Result: The intervention did not achieve statistical significance in the Intention-to-Treat (ITT) analysis.
-
Event Rates: 7.9% in the Treatment arm (24 events) vs. 11.0% in the Standard Care arm (34 events).
-
Context: Although there was a numerical trend favoring the treatment, the lack of statistical significance means the trial failed to prove that vitamin D normalization reduces the overall burden of these combined cardiovascular events.
Key Secondary & Specialized Outcomes
Recurrent Myocardial Infarction (MI):
-
Result: A statistically significant reduction in the risk of a second heart attack.
-
Event Rates: 3.8% (12 events) in the Treatment arm vs. 7.9% (25 events) in the Standard Care arm.
-
Statistical Precision: Log-rank p=0.03.
-
Clinical Impact: This translates to an Absolute Risk Reduction (ARR) of 4.1% and a Number Needed to Treat (NNT) of approximately 24 to prevent one recurrent heart attack over the 4-year follow-up period.
Dosing & Biomarker Response:
-
Baseline Deficiency: 87.0% of participants had vitamin D levels <40 ng/mL at entry.
-
Aggressive Dosing Required: To reach the target (>40 ng/mL), 58.8% of the treatment group required a starting dose of 5,000 IU daily—more than 6 times the standard RDA. This confirms that “standard” doses (e.g., 800 IU) are likely insufficient for normalization in this population.
Harms and Adverse Events:
-
Safety: The study protocol included safety monitoring for hypercalcemia and vitamin D toxicity. The researchers reported observing no adverse outcomes associated with the high-dose supplementation, suggesting the “treat-to-target” range (40-80 ng/mL) is safe under monitoring.
Assertive Critical Appraisal
Risk of Bias (RoB 2 Framework)
Overall Risk of Bias Judgment: Some concerns.
-
Blinding: The trial was open-label. Because the intervention required frequent blood draws and dose adjustments, participants and clinicians knew who was receiving treatment. This introduces performance bias (potential for better general care in the monitored group) and detection bias (potential influence on reporting subjective symptoms), though the “hard” endpoints of death and MI are less susceptible to this.
Composite Endpoints
-
Inconsistency: The discordance between the negative primary composite (MACE) and the positive single component (MI) is a critical limitation. When a composite is negative, “picking out” a positive component is statistically risky (multiplicity). However, the magnitude of the effect on MI (halving the risk) and the biological plausibility (anti-inflammatory effects of Vitamin D) lend some credibility to the finding, provided it is replicated.
Reporting Quality Assessment (CONSORT)
-
Preliminary Data: This analysis is based on a conference abstract and poster figures. Key statistical details often found in full manuscripts, such as Hazard Ratios (HR) with 95% Confidence Intervals (CIs) for the primary outcome, are currently missing.
-
Attrition: The Consort Diagram (Figure 1) indicates 630 patients were randomized. A full assessment of loss-to-follow-up is pending the full manuscript.
Applicability
-
Population: The study is highly specific to post-ACS patients (mostly MI survivors) with documented vitamin D deficiency.
-
Implementation: The intervention is not a simple “take a vitamin” advice; it is a medicalized regimen requiring quarterly testing and dose titration. The high doses used (5,000+ IU) are not standard over-the-counter recommendations.
Research Objective
To determine if a “treat-to-target” strategy of vitamin D supplementation (aiming for serum levels \ge 40 ng/mL) reduces the risk of major adverse cardiovascular events in patients with recent Acute Coronary Syndrome, compared to standard care.
Study Design
-
Design: Parallel-group, Open-label, Randomized Clinical Trial (RCT).
-
Allocation: 1:1 Randomization (314 Treatment vs. 316 Standard Care).
-
Intervention: Personalized D3 dosing adjusted every 3 months to maintain levels between 40-80 ng/mL.
-
Follow-up: Average of 4.2 years (concluded March 2025).
Setting and Participants
-
Setting: Intermountain Health system (Utah, USA).
-
Participants: 630 adults (Mean age 62.6 years; 78.1% Male).
-
Key Inclusion: Diagnosis of ACS; 85% had a prior MI.
-
Key Characteristic: 87% had insufficient vitamin D (<40 ng/mL) at baseline.
Bibliographic Data
Title: A Randomized Clinical Trial Evaluating Vitamin D Normalization on Major Adverse Cardiovascular-Related Events Among Acute Coronary Syndrome Patients: The TARGET-D Trial
Authors Heidi T. May, PhD, MSPH, et al.
Source American Heart Association (AHA) Scientific Sessions 2025
Type Conference Abstract (Abstract 4382525)
Links AHA Newsroom | Intermountain Health
Mandatory Disclaimer: This AI-generated analysis is for informational and research purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of a qualified health provider with any questions you may have regarding a medical condition.
Original Article:
News releases:
Heart attack risk halved in adults with heart disease taking tailored vitamin D doses
