Comment:
Here we have another publication adding to the growing body of evidence supporting Natural Desiccated Thyroid (NDT) for the subset of patients who remain symptomatic on standard levothyroxine monotherapy.
While this article is a “mini-review” and not a large-scale, rigorous RCT, it correctly highlights the crucial clinical reality: patient-reported outcomes and preferences matter. The review gathers important data—from the authors’ own case series to the notable Hoang et al. trial where nearly half of participants preferred NDT—showing that this is a safe and effective option for this select group.
Furthermore, the discussion of a plausible mechanism, such as DIO2 polymorphisms, provides a scientific basis for why L-T4 monotherapy is not a one-size-fits-all solution. This paper serves as yet another reminder that there is a persistent, symptomatic minority and consider NDT a legitimate and valuable therapeutic tool.
Summary:
🩺 CLINICAL BOTTOM LINE
This mini-review argues for considering Natural Desiccated Thyroid (NDT) as a potential option for a small, highly selected group of hypothyroid patients who remain symptomatic despite standard levothyroxine (L-T4) monotherapy. The authors suggest that in this context, patient-reported benefits—even those derived from lower-quality evidence like case series—can be viewed as valid evidence of potential benefit.
However, this article is a mini-review, not a rigorous systematic review. Its conclusions are drawn from a limited and selected body of evidence, including the authors’ own case series, two small RCTs with mixed results, and arguments for biological plausibility. Therefore, its claims should be interpreted with caution.
📄 RESULTS
As a review, this paper summarizes existing literature rather than presenting new pooled data.
-
Persistent Symptoms: The authors highlight that up to 10% of hypothyroid patients treated with levothyroxine report impaired quality of life, even with serum TSH levels in the normal range.
-
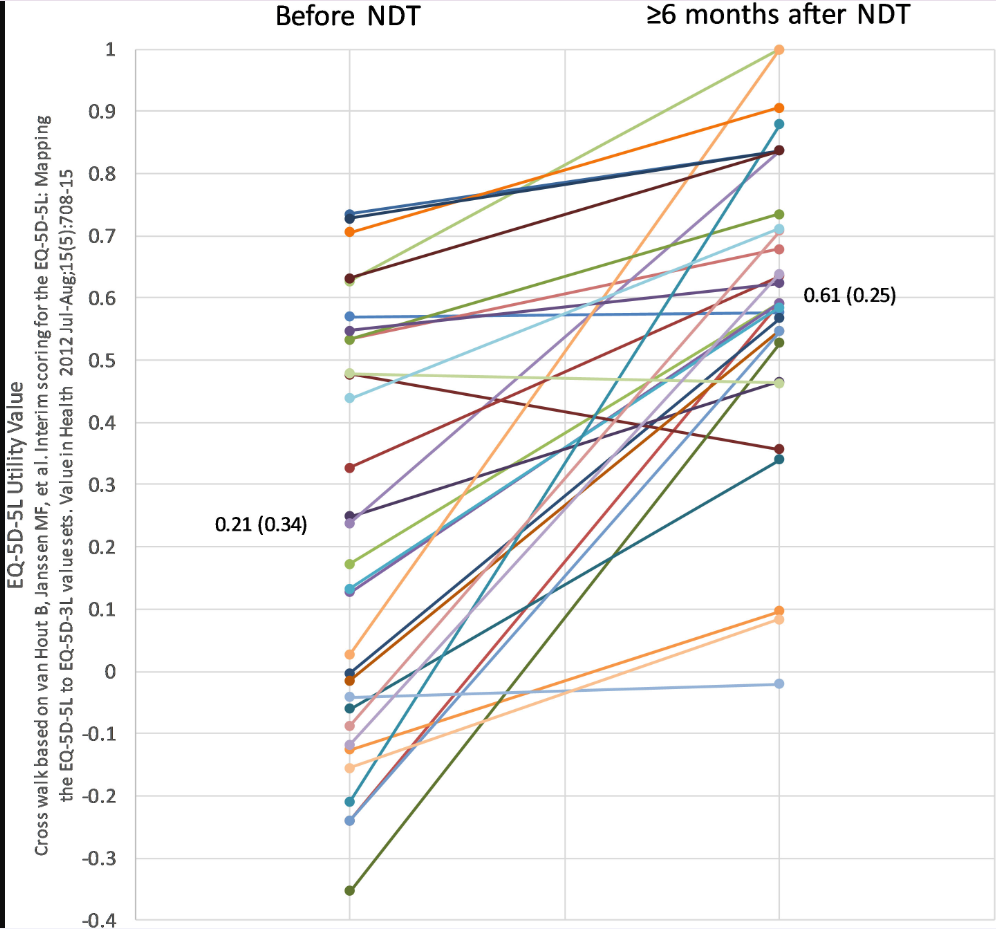
Evidence from Case Series: The review prominently features the authors’ own recent case series, which reported that symptomatically unresponsive patients experienced significant benefit after switching to NDT. This was measured by improvements in quality-of-life scores (EQ-5D-5L and ThyPRO).
-
Evidence from RCTs: The paper discusses two key RCTs:
-
One trial (Hoang et al.) comparing NDT to levothyroxine did not find a significant improvement in quality of life. However, it did find that NDT was associated with modest weight loss (approx. 4 lbs) and that nearly half (48.6%) of participants preferred NDT.
-
A subsequent study by the same group found that the most symptomatic patients showed a strong preference for treatments containing liothyronine (T3), which NDT includes.
-
Potential Mechanism: The authors suggest a pharmacogenetic explanation for why some patients may benefit from NDT (which contains both T4 and T3). Polymorphisms in the deiodinase-2 (DIO2) enzyme, present in about 13% of the population, may reduce the local conversion of T4 to active T3 in tissues like the brain, even if serum levels are normal.
-
Historical Context: The review notes that NDT was the standard of care until the 1970s. It was replaced by levothyroxine monotherapy due to concerns over the potency and stability of NDT preparations and levothyroxine’s favorable pharmacokinetics. The authors contend that potency issues were largely resolved by 1985.
🔬 ASSERTIVE CRITICAL APPRAISAL
-
Certainty of Evidence (GRADE Framework): This article is a “Mini Review” and does not provide a formal GRADE assessment. Based on the evidence it presents (a mix of a case series, mechanistic theories, and small RCTs with mixed primary outcomes), the overall certainty of evidence for NDT’s benefit in this patient group remains Low to Very Low. The authors acknowledge the body of opinion is divided and rightly call for large, well-designed RCTs to clarify which individuals might benefit.
-
Heterogeneity: This is not a meta-analysis, so statistical heterogeneity (I²) is not reported. However, the clinical heterogeneity is high, with conflicting results between the authors’ case series (showing benefit) and the primary endpoint of a key RCT (showing no quality-of-life benefit).
-
Publication Bias: The authors do not perform or discuss an assessment for publication bias. A mini-review is highly susceptible to selection bias, and the authors prominently feature their own prior work, which supports their position.
-
Reporting Quality Assessment (PRISMA): This paper is a narrative mini-review, not a systematic review, and it does not follow PRISMA guidelines.
-
You must note that the paper does not include a PRISMA flow diagram showing how studies were selected.
-
It also fails to describe a systematic search strategy.
-
These omissions are critical. It means this review cannot be assumed to be a balanced or comprehensive summary of all available evidence. It should be read as a perspective or advocacy piece arguing for a specific clinical approach.
🎯 RESEARCH OBJECTIVE
The paper’s implied objective (PICO) is to review the evidence for:
-
P (Population): Patients with primary hypothyroidism who remain symptomatic despite adequate levothyroxine monotherapy.
-
I (Intervention): Natural Desiccated Thyroid (NDT).
-
C (Comparison): Levothyroxine monotherapy (implied).
-
O (Outcome): Symptom alleviation and improvement in quality of life.
DESIGN AND PARTICIPANTS
-
Study Design: This is a “Mini Review,” which is a form of narrative review. It summarizes the authors’ perspective on the topic, supported by a non-systematic selection of literature.
-
Participants: Not applicable, as this is a review. It cites studies involving patients with hypothyroidism.
📚 BIBLIOGRAPHIC DATA
-
Title: Natural desiccated thyroid for the treatment of hypothyroidism?
-
Authors: Heald AH, Taylor P, Premawardhana L, Stedman M and Dayan C
-
Journal: Frontiers in Endocrinology
-
Year: 2024
-
DOI: 10.3389/fendo.2023.1309159
This AI-generated analysis is for informational and research purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of a qualified health provider with any questions you may have regarding a medical condition.
Original Article:
Full text pdf: Natural desiccated thyroid for the treatment of hypothyroidism
Copyright © 2024 Heald, Taylor, Premawardhana, Stedman and Dayan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
