Comment:
This is a well-designed, double-blind, crossover trial putting DTE and levothyroxine head-to-head. The top line findings, that the primary outcomes for the whole group weren’t different, is a great start for substantiating that at the least Natural Desiccated Thyroid is equivalent to levothyroxine.
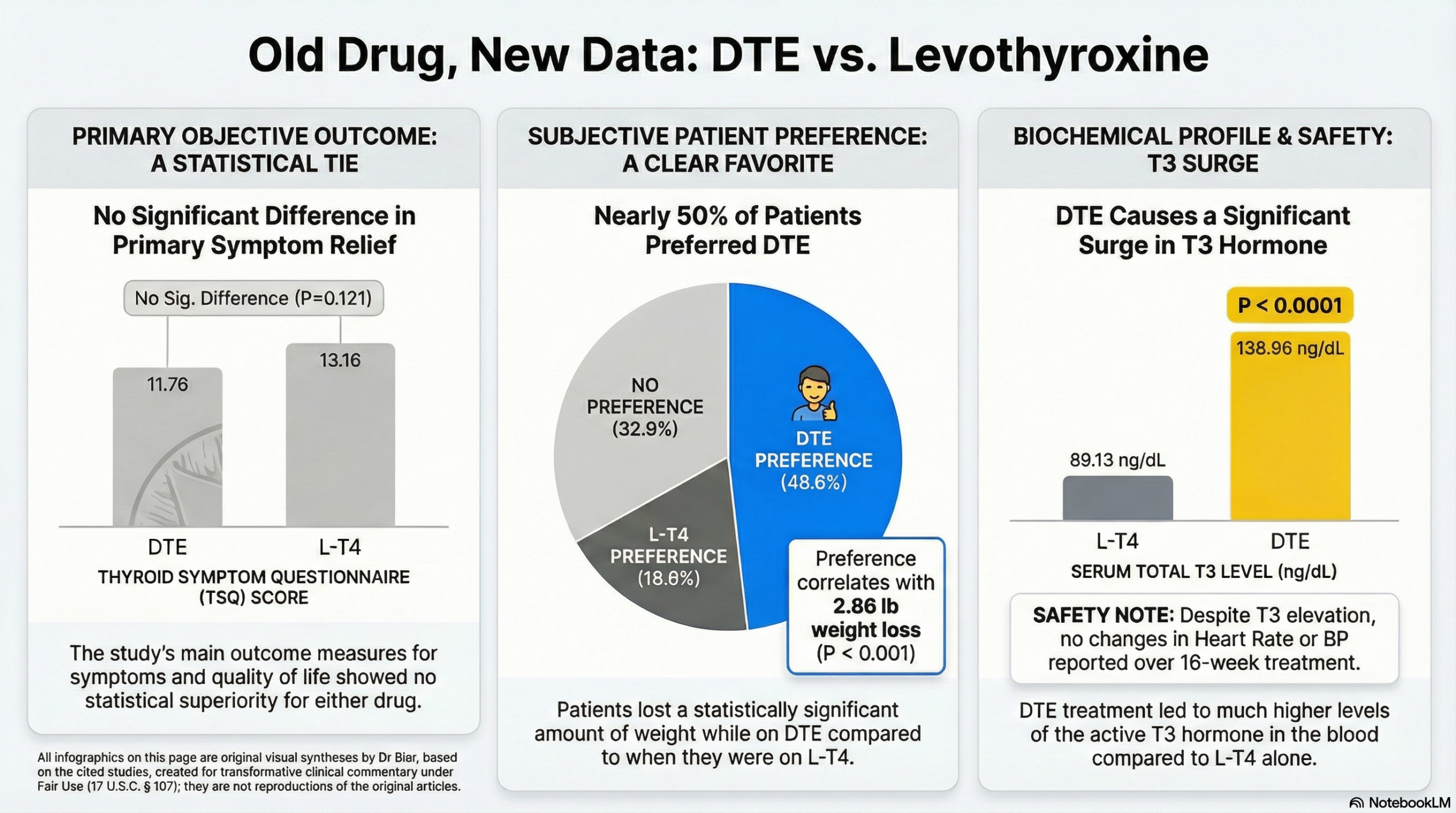
However the secondary findings, although in a sub-group population are very intriguing and underline what we commonly see in clinical practice. Patients lost an average of nearly 3 pounds on DTE and nearly 50% of patients preferred DTE! Even though the subgroup analysis needs to be interpreted with caution, it strongly suggests that for a certain subset of patients, likely those that don’t convert the synthetic T4 over to the more active T3 form, DTE may offer real, subjective benefits in symptoms and well-being that L-T4 monotherapy just doesn’t provide.
Summary:
🩺 Clinical Bottom Line
This small but well-designed, double-blind, crossover trial found that desiccated thyroid extract (DTE) was not superior to levothyroxine (L-T4) for improving hypothyroid symptoms or neurocognitive function in the overall study group. However, DTE treatment did result in a modest but statistically significant average weight loss of approximately 3 pounds compared to L-T4. The most notable finding was that nearly half (48.6%) of the patients preferred DTE over L-T4, compared to only 18.6% who preferred L-T4. While a subgroup analysis of the DTE-preferring patients did show significant symptom improvement, this finding should be viewed with extreme caution as it is a post-hoc analysis highly susceptible to bias.
📊 Results in Context
-
Primary Outcome: The study’s primary outcome measures included symptom scores (Thyroid Symptom Questionnaire [TSQ]), general well-being (General Health Questionnaire [GHQ]-12), and a battery of neurocognitive tests.
-
Across all 70 participants, there were no statistically significant differences in hypothyroid symptoms, quality of life, or neurocognitive measurements between the DTE and L-T4 treatment periods.
-
-
Key Secondary & Patient-Reported Outcomes (PROs):
-
Patient Preference: This was the study’s most significant finding. When asked at the end of the trial, 34 patients (48.6%) preferred DTE, 13 (18.6%) preferred L-T4, and 23 (32.9%) had no preference. The preference for DTE was statistically significant.
-
Weight Loss: Patients weighed, on average, 2.86 pounds less while on DTE therapy compared to L-T4 therapy (172.9 lbs vs. 175.7 lbs; P < .001).
-
Biochemical Profile: To maintain the target TSH (0.5-3.0 µIU/mL), the two therapies resulted in different biochemical profiles. During DTE treatment, serum total T3 was significantly higher and serum free T4 was significantly lower than during L-T4 treatment. Serum TSH was also slightly, but significantly, higher in the DTE group (1.67 vs. 1.30 µIU/mL).
-
PROs (Subgroup): In the subgroup of 34 patients who stated a preference for DTE, they showed significant improvements in subjective symptoms on both the TSQ and GHQ-12 questionnaires while taking DTE (P < .001 for both).
-
-
Harms and Adverse Events:
-
No adverse effects were reported with either treatment.
-
All patients tolerated both DTE and L-T4 equally well. There were no significant changes in heart rate or blood pressure between the two groups.
-
🧐 Assertive Critical Appraisal
-
Risk of Bias (RoB 2 Framework):
-
Overall judgment: Some concerns.
-
Critique: The randomized, double-blind, crossover design is a significant strength, as patients serve as their own controls, minimizing confounding. Randomization and allocation concealment appear robust.
-
The primary concern arises from the interpretation of the results. The study’s pre-specified primary outcomes (symptoms and neurocognition in the full group) were negative. The positive findings for symptom improvement were only identified in a subgroup analysis conducted after unblinding and assessing patient preference. This introduces a major risk of bias, as the subgroup is defined by the very outcome (preference) it seeks to explain.
-
-
Subgroup Analyses:
-
The study’s most-discussed finding—that some patients “do better” on DTE—is based entirely on a subgroup analysis of those who preferred DTE.
-
As per standard appraisal, subgroup claims are often unreliable. While this analysis was pre-specified, it is not a randomized comparison. It is plausible that the patients who felt better (for any number of reasons, including the modest weight loss) subsequently stated a preference, making this a circular finding. These results should be considered hypothesis-generating, not conclusive.
-
-
Appraisal of Patient-Reported Outcomes (CONSORT-PRO):
-
The PROs (TSQ, GHQ-12, BDI) were appropriately pre-specified as primary outcome measures.
-
The dropout rate was low (10%), meaning missing PRO data was unlikely to have biased the main (negative) finding. The study’s conclusion that DTE offered no significant PRO benefit overall is sound.
-
-
Reporting Quality Assessment (CONSORT):
-
The reporting quality is high. The paper includes a CONSORT flow diagram (Figure 1) detailing participant allocation and follow-up.
-
It also clearly describes the methods for randomization (computer-generated list) and allocation concealment (maintained by a physician not involved in the study), which strengthens confidence in the trial’s validity.
-
-
Applicability:
-
The study population (patients with primary hypothyroidism on stable L-T4) is directly relevant to general clinical practice.
-
The findings do not support switching all patients to DTE. However, they do provide evidence that for a patient who is dissatisfied with L-T4 monotherapy, a trial of DTE is a reasonable consideration. It may offer a modest benefit in weight and subjective well-being for some, with no apparent short-term adverse effects.
-
📝 Study Details
-
Research Objective:
-
To investigate the effectiveness of DTE compared with L-T4 on symptoms, cognitive function, and general well-being in hypothyroid patients.
-
-
Study Design:
-
A randomized, double-blind, crossover study.
-
Participants were randomized to one of two sequences: (1) DTE for 16 weeks, then L-T4 for 16 weeks, or (2) L-T4 for 16 weeks, then DTE for 16 weeks.
-
A total of 85 patients were assessed for eligibility, 78 were randomized, and 70 completed both study periods.
-
-
Setting and Participants:
-
Setting: A single tertiary care center (Walter Reed National Military Medical Center).
-
Participants: 70 patients (age 18-65) with primary hypothyroidism who had been on a stable dose of L-T4 for at least 6 months prior to enrollment.
-
Exclusion criteria included pregnancy, coronary artery disease, significant lung/renal/liver disease, active cancer, and use of medications known to interfere with thyroid hormone (e.g., amiodarone, proton pump inhibitors, iron).
-
📚 Bibliographic Data
-
Title: Desiccated Thyroid Extract Compared With Levothyroxine in the Treatment of Hypothyroidism: A Randomized, Double-Blind, Crossover Study
-
Authors: Thanh D. Hoang, Cara H. Olsen, Vinh Q. Mai, Patrick W. Clyde, and Mohamed K. M. Shakir
-
Journal: The Journal of Clinical Endocrinology and Metabolism (J Clin Endocrinol Metab)
-
Year: 2013
-
DOI: 10.1210/jc.2012-4107
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
