Regulatory Transition and Clinical Continuity of Desiccated Thyroid Extract: A Strategic Analysis of FDA Policy Evolutions 2025–2026
Regulatory History: The Distinction Between Grandfathered and GRASE Status
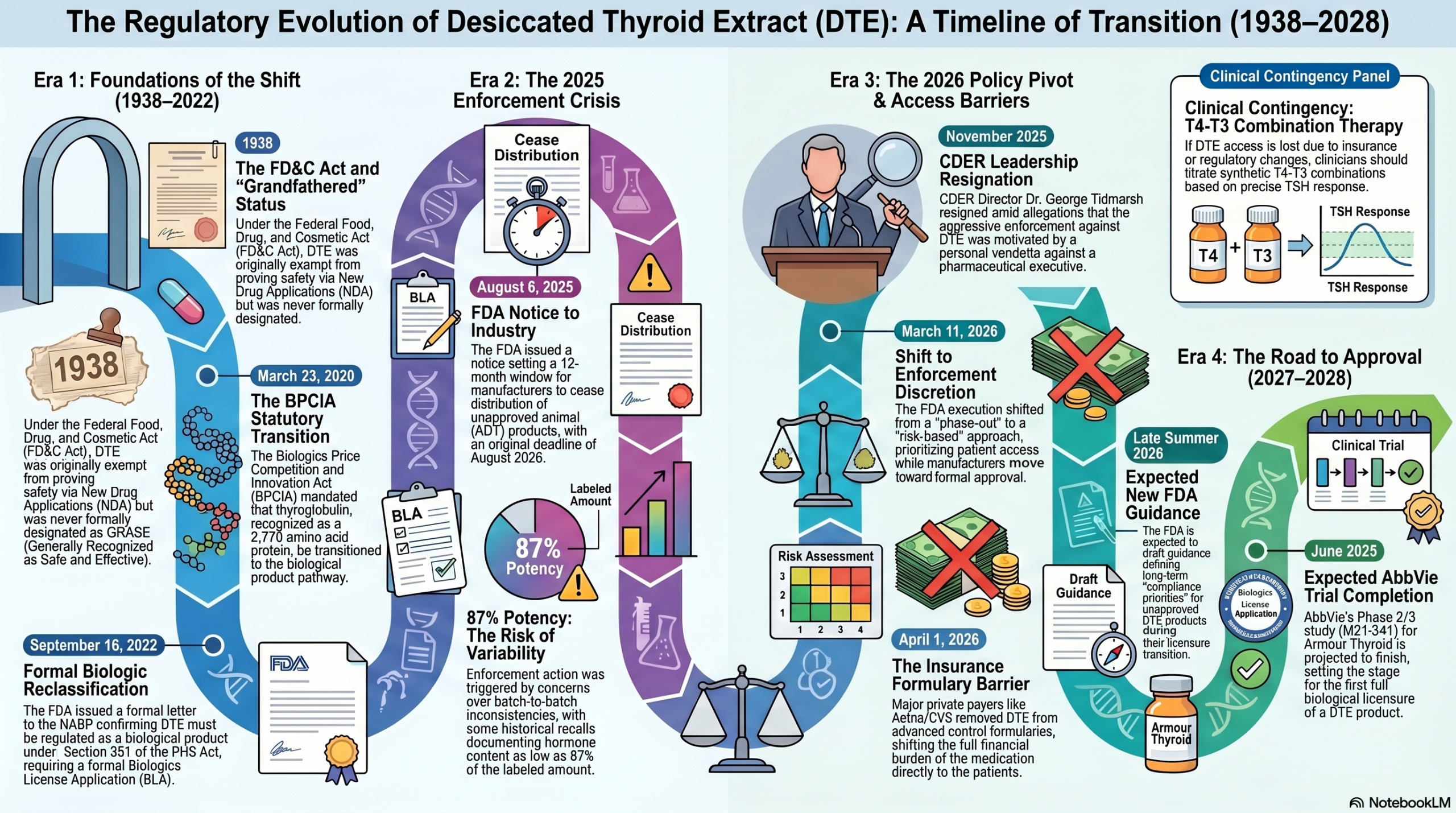
Desiccated Thyroid Extract (DTE) has been utilized for thyroid hormone replacement since the late 19th century, pre-dating the formal regulatory framework of the U.S. Food and Drug Administration (FDA). For decades, DTE occupied a regulatory space often referred to as “grandfathered.” Under the Federal Food, Drug, and Cosmetic Act (FD&C Act) of 1938, drugs marketed prior to the Act’s inception were technically exempt from the requirement to prove safety through a New Drug Application (NDA), provided their labeling and use remained unchanged.
It is critical for clinicians to distinguish “grandfathered” status from “Generally Recognized as Safe and Effective” (GRASE) status. DTE products were never formally designated as GRASE by the FDA; rather, they remained on the market through a historical lack of active enforcement. Unlike synthetic levothyroxine (T4), which eventually underwent formal FDA review and approval (around 2000), DTE preparations were historically managed by manufacturers according to United States Pharmacopeia (USP) monographs without direct FDA pre-market oversight of their safety or efficacy.
The transition to the current “unapproved biologic” status was a consequence of the Biologics Price Competition and Innovation Act (BPCIA) of 2009, which mandated that certain protein products previously regulated as drugs transition to the biological product pathway with a statutory transition date of March 23, 2020. This shift was formalized for this product class in a September 16, 2022, letter from the FDA to the National Association of Boards of Pharmacy (NABP).1 The agency determined that because DTE contains thyroglobulin—a protein of 2,770 amino acids—it meets the statutory definition of a “protein” under 21 CFR § 600.3 and must be regulated as a biological product under Section 351 of the Public Health Service (PHS) Act.1 Because the PHS Act does not contain a grandfathering provision, DTE products must now be marketed through an approved Biologics License Application (BLA).
The August 2025 Enforcement Notice: Potency and Safety Concerns
In August 2025, the FDA issued formal notifications expressing its intent to take enforcement action against unapproved animal-derived thyroid (ADT) products.2 The agency issued a formal Notice to Industry on August 6, 2025, which highlighted several safety concerns as the basis for this shift:
- Adverse Event Data: The FDA stated it was aware of more than 500 adverse event reports associated with ADT products recorded between 1968 and February 2025.2
- Potency Variability: The agency raised concerns regarding batch-to-batch inconsistencies. Historical enforcement data, such as the September 3, 2020, voluntary recall of Nature-Throid and WP Thyroid, documented lots containing as low as 87% of the labeled hormone content.3
- Clinical Sensitivity: Given the narrow therapeutic index of thyroid hormones, the agency argued that even minor fluctuations in potency could lead to clinically significant hypo- or hyperthyroidism, especially in vulnerable populations such as pregnant women.2
The initial notice suggested a 12-month window for manufacturers to cease distribution, which was interpreted by many as a hard deadline for August 2026.
The Tidmarsh Allegations and Ethical Oversight
The 2025 enforcement actions were complicated by allegations of misconduct involving Dr. George Tidmarsh, who served as Director of the Center for Drug Evaluation and Research (CDER) starting in July 2025. Dr. Tidmarsh resigned in November 2025 amid an investigation by the Department of Health and Human Services (HHS) into “serious concerns about his personal conduct”.4
According to a lawsuit filed by Aurinia Pharmaceuticals (Aurinia Pharmaceuticals Inc. v. Tidmarsh, Case No. 8:2025cv03593) 5, Dr. Tidmarsh allegedly used his official position to pursue a personal vendetta against pharmaceutical executive Kevin Tang. The lawsuit and subsequent congressional inquiries6 allege that Dr. Tidmarsh pushed for the removal of DTE to target companies associated with Mr. Tang, including American Laboratories, a primary supplier of DTE active ingredients. While these allegations provided a political catalyst for re-evaluating the “ban,” the FDA has maintained that its underlying scientific concerns regarding unapproved biologics remain consistent with its long-term regulatory framework. This stance aligns with the FDA’s broader Unapproved Drugs Initiative launched in 2006.
The March 2026 Policy Clarification: A Shift to Enforcement Discretion
On March 11, 2026, the FDA issued a follow-up communication that clarified its enforcement posture, effectively moving the agency from a “phase-out” strategy to a “risk-based” monitoring approach.7
Official communications confirm the following:
- Commitment to Access: The agency stated it is “committed to ensuring safety of patients currently using unapproved animal-derived thyroid medications while manufacturers are working toward FDA approval”.8
- Enforcement Priority: The FDA will prioritize enforcement against products that “pose the highest risk to public health,” such as those failing quality standards, rather than a blanket removal of all DTE products by August 2026.
- Future Guidance: The FDA intends to issue draft guidance before August 2026 describing the “conditions of continued enforcement discretion” for marketed unapproved DTE.7
Manufacturer Progress and Clinical Trial Status
The long-term availability of DTE depends on manufacturers successfully navigating the BLA process. Current efforts include:
- Acella Pharmaceuticals / Neuvosyn Laboratories: In September 2025, the manufacturer announced positive topline results from a Phase 2 trial (NCT05712421) for “North Star,” an investigational DTE product.9 According to manufacturer announcements, the study defined a safe dose-conversion ratio from levothyroxine to North Star. Clinicians should note these results were released via corporate announcement and have not yet undergone formal peer review.
- AbbVie (Armour Thyroid): As of early 2026, AbbVie is actively Recruiting for a Phase 2/3 clinical trial (Study M21-341, NCT06345339) to assess the safety and efficacy of oral Armour Thyroid compared to synthetic T4.10 The estimated primary completion date is September 2027, with full study completion expected in June 2028.
Given that clinical trials are projected to continue through 2027, the period of “enforcement discretion” will likely need to extend for several years beyond the original 2026 window to allow for trial completion and FDA review.
Clinical Context and Society Guidelines
The American Thyroid Association (ATA) and the American Association of Clinical Endocrinology (AACE) continue to recommend synthetic levothyroxine (T4) monotherapy as the standard first-line treatment for hypothyroidism due to its stable dosing and safety profile. However, the ATA acknowledges that while it is not recommended as first-line therapy, “it is the treatment of choice for some patients” and supports the principle of “personalized patient therapy”.11
Formulary Changes and Access Barriers
While regulatory pressure from the FDA has eased, private sector barriers have intensified. A January 30, 2026, member notice from Aetna/CVS informed patients that DTE medications would be removed from standard commercial “Advanced Control” formularies effective April 1, 2026.12
|
Barrier Type |
Access Impact |
Current Status |
|
Formulary Exclusion |
High out-of-pocket costs (Approx. $27–$144/month*) |
Effective April 1, 202612 |
|
Regulatory Status |
Enforcement discretion (Continued access) |
Conditional on BLA progress7,8 |
|
Compounding |
Prohibited by regulation as a biologic |
Not eligible per FDA8 |
*Approximate retail cash prices based on pharmacy data as of May 2026
Prognosis for 2026 and Beyond
The 1.5 million Americans currently using DTE are unlikely to lose access in August 2026. The FDA’s pivot to “risk-based enforcement” provides a structured pathway for products to remain available while clinical trials proceed.
Clinical Contingency Planning: In the event that a BLA is ultimately denied or DTE products are removed, medical literature suggests that levothyroxine-liothyronine (T4-T3) combination therapy is the closest pharmacologic alternative. DTE typically contains a T4-T3 ratio of approximately 4:1 by weight (roughly 38 mcg T4 and 9 mcg T3 per grain), which is a supraphysiologic amount of T3 relative to normal human thyroid secretion. Clinicians can titrate combination therapy based on TSH and clinical response to approximate this ratio while utilizing approved, standardized synthetic products.
Stakeholders should monitor the FDA’s website for the promised draft guidance on “compliance priorities,” expected in late summer 2026, which will define the long-term rules for the DTE market as it transitions toward full biologic licensure.
Works Cited
- FDA Letter to NABP Regarding DTE Biologic Reclassification (September 16, 2022).(https://join.a4pc.org/hubfs/PDFs/MEMO-EO-FDA-Letter-re-DTE5.pdf)
- FDA Notice to Industry: Animal-Derived Thyroid Products (August 6, 2025). https://www.fda.gov/media/188081/download
- FDA: RLC Labs Nature-Throid Voluntary Recall Notice (September 3, 2020). https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/rlc-labs-inc-issues-voluntary-nationwide-recall-all-lots-nature-throidr-and-wp-thyroidr-current
- HHS Statement on Resignation of CDER Director George Tidmarsh (November 2, 2025). (On file/available via major news outlets).
- U.S. District Court for the District of Maryland: Aurinia Pharmaceuticals Inc. v. Tidmarsh, Case No. 8:2025cv03593. https://dockets.justia.com/docket/maryland/mddce/8:2025cv03593/593827
- Congressional Correspondence: Representative Abe Hamadeh Inquiry to FDA (November 4, 2025).(https://hamadeh.house.gov/news/documentsingle.aspx?DocumentID=470)
- FDA Official Letter to Manufacturers Regarding Risk-Based Enforcement (March 11, 2026). https://www.fda.gov/media/191621/download
- FDA Official Guidance Page: “Actions to Address Unapproved Thyroid Medications”. https://www.fda.gov/drugs/enforcement-activities-fda/fdas-actions-address-unapproved-thyroid-medications
- Acella/NP Thyroid: Product Updates & Clinical Trial Success Press Release (September 23, 2025). https://npthyroid.com/faq/product-updates/
- ClinicalTrials.gov. AbbVie Armour Thyroid Phase 2/3 Study (NCT06345339).(https://clinicaltrials.gov/study/NCT06345339)
- American Thyroid Association Statement on Desiccated Thyroid Extract (September 18, 2025). https://www.thyroid.org/ata-statement-desiccated-thyroid-extract/
- Aetna/CVS Member Notice: Commercial Template Formulary Changes (January 30, 2026).(https://www.aetna.com/content/dam/aetna/pdfs/aetnacom/individuals-families-health-insurance/document-library/pharmacy/SOC-for-04.01.26-Advanced-Control.pdf)
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
