Comment:
I have used PRP for thumb joint pain and seen it been very helpful. This trial paints a very hopeful picture for the use of PRP for thumb CMC OA treatments. While we definitely need larger controlled trials to confirm these results, the initial data—specifically the high rate of moderate-to-excellent improvement—indicates that PRP could soon fill a critical gap for patients seeking effective, long-term non-surgical relief.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
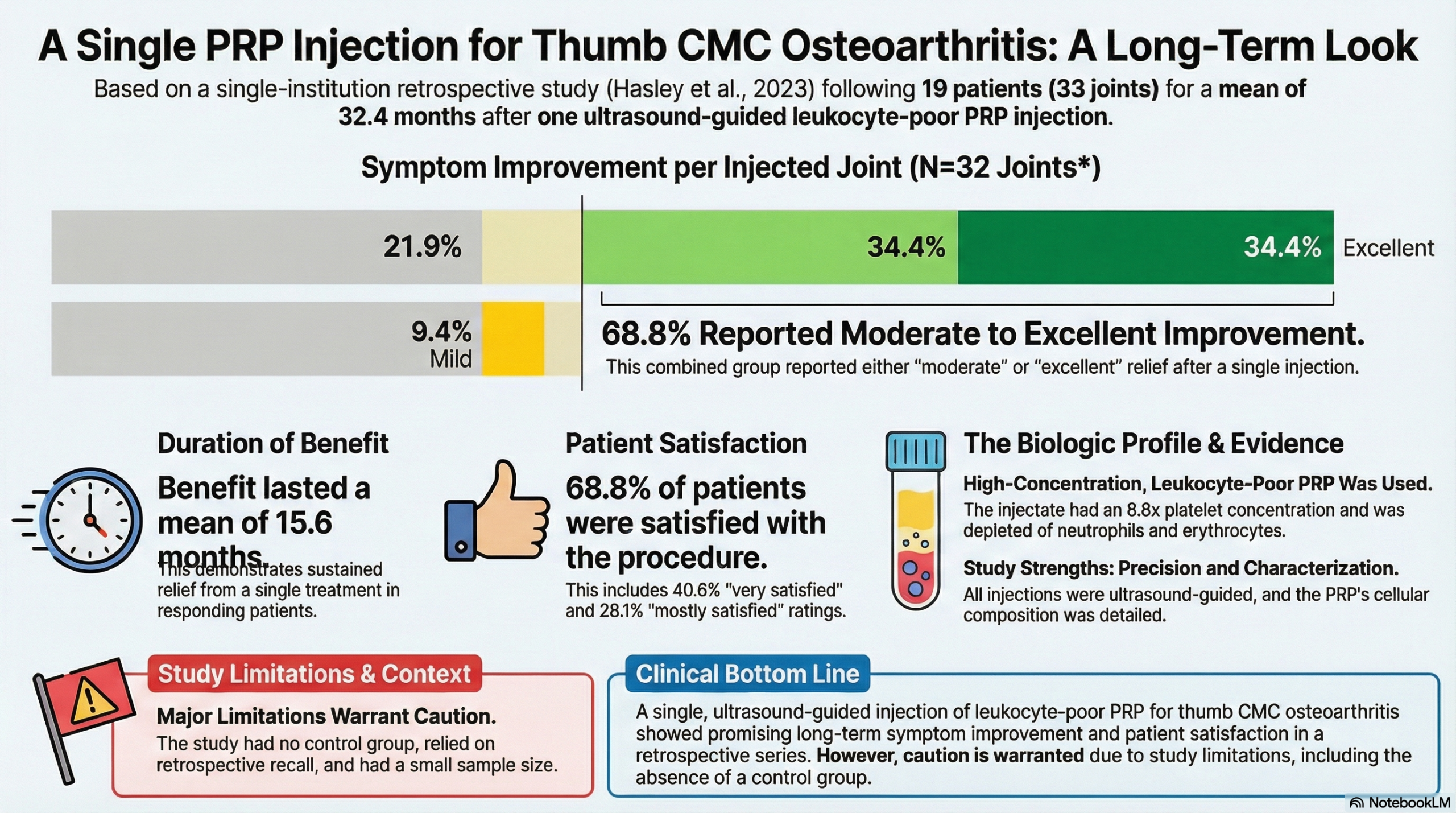
This retrospective observational study suggests a strong association between a single, ultrasound-guided PRP injection (specifically, one that is depleted in erythrocytes and neutrophils but enriched in lymphocytes and monocytes) and worthwhile pain relief for patients with thumb CMC OA. In a cohort of 19 patients (33 joints), 68.8% of injected joints were rated as having moderate or excellent symptom improvement, with a high patient satisfaction rate (68.8% mostly or very satisfied). The mean patient-reported duration of benefit was relatively long at 15.6 months (\pm19.5). However, as an observational study, it can only identify associations, not prove that the PRP injection caused the reported improvement, as there was no control or comparison group, and the patient-reported outcomes relied heavily on potentially biased post-hoc telephone surveys.
Results in Context
Main Results
The study evaluated 33 thumb CMC joints from 19 adult patients (average age 65.0 years).
- Symptom Improvement: 68.8% of injected joints were reported as having moderate or excellent symptom improvement.
- 34.4% rated improvement as moderate.
- 34.4% rated improvement as excellent.
- 21.9% reported no improvement.
- Duration of Benefit: The mean patient-reported duration of benefit was 15.6 months (\pm19.5) months, with a mean follow-up duration of 32.4 months.
- Satisfaction: 68.8% of procedures were rated as mostly or very satisfied.
- 40.6% were very satisfied and 28.1% were mostly satisfied.
- 18.8% were very unsatisfied.
- PRP Cellular Composition: The PRP product used had a mean platelet concentration factor of 8.80 (\pm4.19) times baseline. The resulting PRP was depleted in erythrocytes (concentration factor 0.02) and neutrophils (concentration factor 0.14), but enriched in lymphocytes (concentration factor 3.76) and monocytes (concentration factor 3.29).
Participants
- Sample Size: 19 adult patients (9 women, 10 men; average age 65.0 years). This resulted in 33 injected thumb CMC joints.
- OA Severity (Eaton Classification): The injected joints were heterogeneous, with most being moderate to severe.
- Grade II: 21.2% (7 joints).
- Grade III: 54.6% (18 joints).
- Grade IV: 24.2% (8 joints).
Assertive Critical Appraisal
Limitations & Bias (STROBE Framework)
The study design, a retrospective case series with a small sample, is the primary methodological limitation and introduces significant risks of bias.
- Lack of Control Group: The absence of a comparison group (e.g., placebo, corticosteroid) makes it impossible to distinguish the true treatment effect from the placebo effect or the natural course of the disease. The positive outcomes reported could be entirely due to patient expectation or regression to the mean.
- Recall Bias: The low response rate to standardized electronic questionnaires (Visual Analog Scale/Quick Disabilities of Arm, Shoulder, and Hand) forced the researchers to obtain most follow-up data via a post-hoc telephone survey. This process introduces significant recall bias as patients are asked to rate past symptom improvement and duration of benefit, potentially inflating positive results.
- Small Sample Size: The sample of 19 patients (33 joints) is insufficient to draw definitive conclusions about efficacy or to determine a relationship between the PRP’s cellular composition and patient outcomes.
Reporting Quality Assessment (STROBE)
The study adequately describes the cohort and the intervention technique, but the inherent nature of the design prevents it from fully addressing confounding.
- Confounding: As an uncontrolled observational study, confounding is the most critical unaddressed challenge. While the paper describes the patient population (age, sex, OA grade), it cannot account for unmeasured confounding variables (e.g., patient motivation, compliance with post-injection activity modification, unrecorded co-interventions) that may have influenced the patient-reported outcomes.
- Selection Bias: The study only included patients who received a PRP injection, which introduces a selection bias because these patients likely had a strong preference or specific clinical profile that led them to choose the PRP procedure over other common options.
Applicability
The findings are applicable to a general clinical practice that utilizes image-guided injections for thumb CMC OA.
- Relevant Procedure: All injections were performed under ultrasound guidance, which has been shown to be more accurate than blind injection for hand joints, making the positive results relevant to contemporary practice.
- Specific PRP Type: The study provides a precise characterization of the cellular composition of the high-platelet, erythrocyte/neutrophil-poor PRP used, which is valuable for comparison with future studies or different commercial kits.
Research Objective
The primary aims were to (1) assess outcomes of PRP injection in patients with thumb CMC OA and (2) characterize the cellular composition of the PRP injected. The secondary aim was to describe the procedural technique used.
Study Design
- Design: Single institution retrospective case series study with chart review and follow-up questionnaires/surveys.
- Setting: Outpatient clinic at a single tertiary care hospital between 2015 and 2020.
- Intervention: Single Platelet-Rich Plasma (PRP) injection into the thumb CMC joint.
- Follow-up: Mean time from injection to telephone survey was 32.4 months (±18.1).
Setting and Participants
- Setting: Physical medicine and rehabilitation, orthopedic surgery hand, and sports medicine clinics at a single institution.
- Participants: 19 adult patients with thumb CMC OA who received a PRP injection.
- Inclusion Criteria: Patients with thumb CMC OA who received a PRP injection.
- Exclusion Criteria: 100 patients with a history of thumb CMC OA were excluded because they received PRP injections in body regions other than the first CMC joint.
Bibliographic Data
| Field | Detail |
|---|---|
| Title | Platelet-Rich Plasma Injection for Thumb Carpometacarpal Joint Osteoarthritis |
| Authors | Ike B. Hasley, MD, Michael M. Bies, DO, John H. Hollman, PT, PhD, Karina Gonzales Carta, MD, Jacob L. Sellon, MD, Jeffrey S. Brault, DO |
| Journal | Archives of Rehabilitation Research and Clinical Translation |
| Year | 2023 |
| DOI | 10.1016/j.arrct.2023.100257 |
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
