Comment:
Starting with the clear caveats that this is a preclinical mouse study and a pre-print (has not undergone peer review yet), this is still a fascinating study.
We typically categorize Vitamin B12 deficiency as a hematologic or neurological issue, often overlooking or underestimating its structural role in muscle bioenergetics. This study brings the musculoskeletal role into greater clarity, identifying a distinct “toxicity of omission”: without adequate B12, the mitochondrial DNA polymerase is forced to substitute uracil for thymine, literally building instability into the mitochondrial genome of skeletal muscle. This is not merely a cofactor shortage that impacts new muscle, but a compromise of the genetic integrity of the muscle that directly stifles oxidative phosphorylation. For the Naturopathic clinician, this provides a mechanistic understanding for what we’ve known, which is there is a link between low B12 status and fatigue and muscle weakness.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
This preclinical study in mice provides mechanistic evidence that Vitamin B12 deficiency directly impairs mitochondrial function in skeletal muscle by causing uracil misincorporation into mitochondrial DNA (mtDNA). While this is a basic science report (preprint, not yet peer-reviewed), it offers a potential biological explanation for the muscle weakness and frailty often observed in B12-deficient older adults. Notably, intramuscular B12 supplementation in aged mice successfully restored specific mitochondrial respiratory activity, suggesting a reversible component to age-related mitochondrial decline.
Results in Context
Mechanism of Action: The One-Carbon Cycle
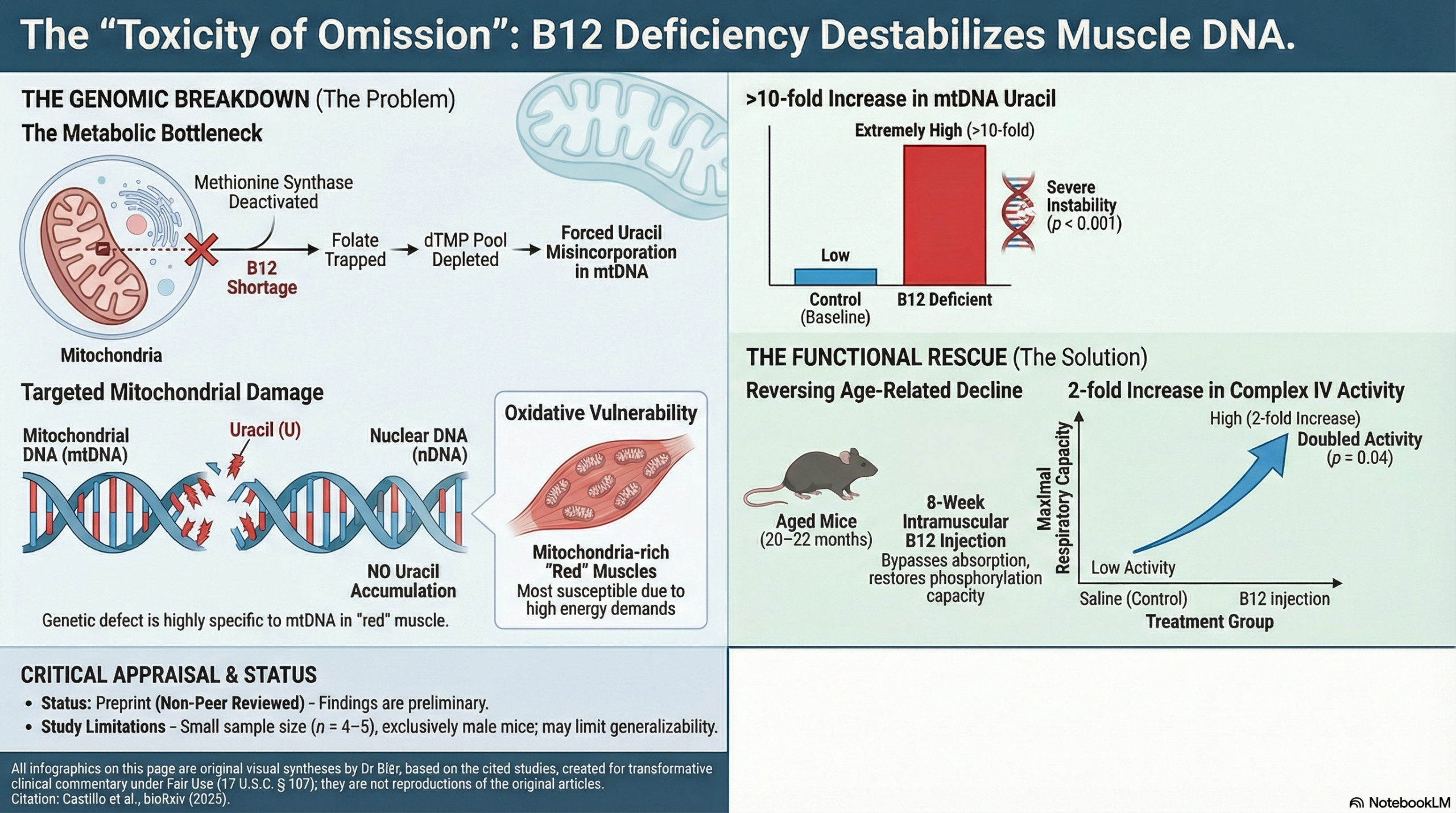
To understand the results, it is necessary to visualize the metabolic bottleneck caused by B12 deficiency. B12 is a cofactor for methionine synthase (MTR). When MTR activity is low (due to lack of B12), folate becomes “trapped” as 5-methyl-THF. This depletes the folate pool required to synthesize thymidylate (dTMP)—the “T” in DNA.
Consequently, the cell is forced to substitute uracil (“U”) for thymine during DNA synthesis, leading to DNA instability. This study confirms this mechanism occurs specifically within the mitochondria of muscle cells.
Primary Findings: Mitochondrial Function & Integrity
- Respiratory Capacity (Tibialis Anterior – Glycolytic Muscle): In mice with genetic B12 deficiency (Mtr^{+/-}), maximal activity was significantly reduced in Complex I (p=0.01), Complex II (p=0.001), and Complex IV (p=0.006) of the electron transport chain.
- Respiratory Capacity (Red Muscle – Oxidative): Dietary B12 deficiency specifically lowered the maximal capacity of Complex I (p=0.02) and Complex IV (p=0.03) in mitochondria-rich red muscle.
- mtDNA Integrity: Mice on a B12-deficient diet exhibited a ~10-fold increase in uracil accumulation in the mtDNA of red muscle (p=0.04) and gastrocnemius (p<0.001).
- Note: This accumulation was specific to mitochondrial DNA; no significant uracil accumulation was found in the nuclear DNA of these muscle tissues.
Intervention in Aged Mice
- Setup: Aged male mice (20-22 months old) received weekly intramuscular B12 injections or saline for 8 weeks.
- Outcome: The B12-supplemented group showed a 2-fold increase in maximal Complex IV activity in the gastrocnemius muscle compared to controls (p=0.04).
Assertive Critical Appraisal
Evidence Level & Applicability
- Preclinical Model: This is an animal study using specific genetic Mtr+/- and dietary manipulations. While the findings are biologically plausible, mouse metabolism and muscle physiology differ from humans. The “B12 deficient” diet was severe (0 mcg g/kg), designed to induce deficiency rapidly.
- Preprint Status: This manuscript is a preprint and has not been certified by peer review. Its findings should be treated as preliminary data rather than established clinical fact.
Study Strengths
- Mechanistic Clarity: The study effectively links the biochemical defect (impaired dTMP synthesis) to a structural pathology (uracil in mtDNA) and a functional deficit (reduced respiration).
- Relevance of Intervention: The use of intramuscular injections in the aged mouse model mimics a common route of administration in clinical geriatrics, increasing the translational interest of the findings.
Limitations & Methodological Concerns
- Small Sample Sizes: Several key experiments relied on very small sample sizes (n=3 to n=6 per group). Small samples increase the risk of Type I errors (false positives) and Type II errors (missing real effects), particularly in variable biological assays like Western blots and respirometry.
- Sex Bias: The study exclusively used male mice. Given that sarcopenia and mitochondrial aging can manifest differently between sexes, the exclusion of female mice limits the generalizability of the findings.
- Inconsistent Genotype Effects: While the dietary deficiency consistently increased mtDNA uracil, the genetic defect Mtr+/- did not reach statistical significance for this specific marker in muscle, despite reducing respiratory capacity. The authors suggest this may be due to the limited sample size.
Reporting Quality Assessment (ARRIVE/biomedical standards)
The reporting is generally clear regarding methods, but the reliance on “representative” blots and low-n quantification without power calculations is a weakness typical of early preclinical reports. The distinction between “Red Muscle” (oxidative) and “Tibialis Anterior” (glycolytic) is well-justified physiologically, adding nuance to the results.
Research Objective
The study aimed to determine if functional B12 deficiency (genetic or dietary) compromises mitochondrial DNA integrity and function in skeletal muscle, and whether B12 supplementation could reverse mitochondrial deficits in aged mice.
Study Design
- Design: Controlled laboratory experiment with 2×2 factorial design (Genotype x Diet) and a separate arm for aging intervention.
- Models: Mtr+/+ vs Mtr+/- mice; Young vs Aged (C57BL6/N) mice.
- Interventions: B12 deficient diet vs Control diet; IM B12 injections vs Saline.
Setting and Participants
- Setting: Laboratory animal facility (Division of Nutritional Sciences, Cornell University).
- Subjects: Male mice (weaned offspring for genetic study; 20-22 month old males for aging study).
Bibliographic Data
- Title: Vitamin B12 supports skeletal muscle oxidative phosphorylation capacity in male mice
- Authors: Castillo LF, Heyden KE, Williamson AR, et al.
- Source: bioRxiv (Preprint)
- DOI: https://doi.org/10.1101/2025.05.19.654973
- Date: Posted August 30, 2025
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
