Comment:
This is a really important study, as it is a randomized controlled trial, vs some of the other retrospective ones. And it is showing some of the same negative outcomes, strengthening the findings of those weaker trial designs.
We are beginning to see a consistent, sobering pattern in the literature regarding Time-Restricted Eating (TRE). This randomized clinical trial adds another layer to the “hierarchy of harm” we have been tracking: the risk that the weight lost during fasting is not the weight we want patients to lose. The core tension revealed here is between the promise of “metabolic hacking” and the physiological reality of catabolism. While proponents of these protocols frequently cite increased growth hormone during fasting as a muscle-sparing mechanism, the hard data from this study suggests that for the average patient, biology prioritizes energy conservation over muscle preservation.
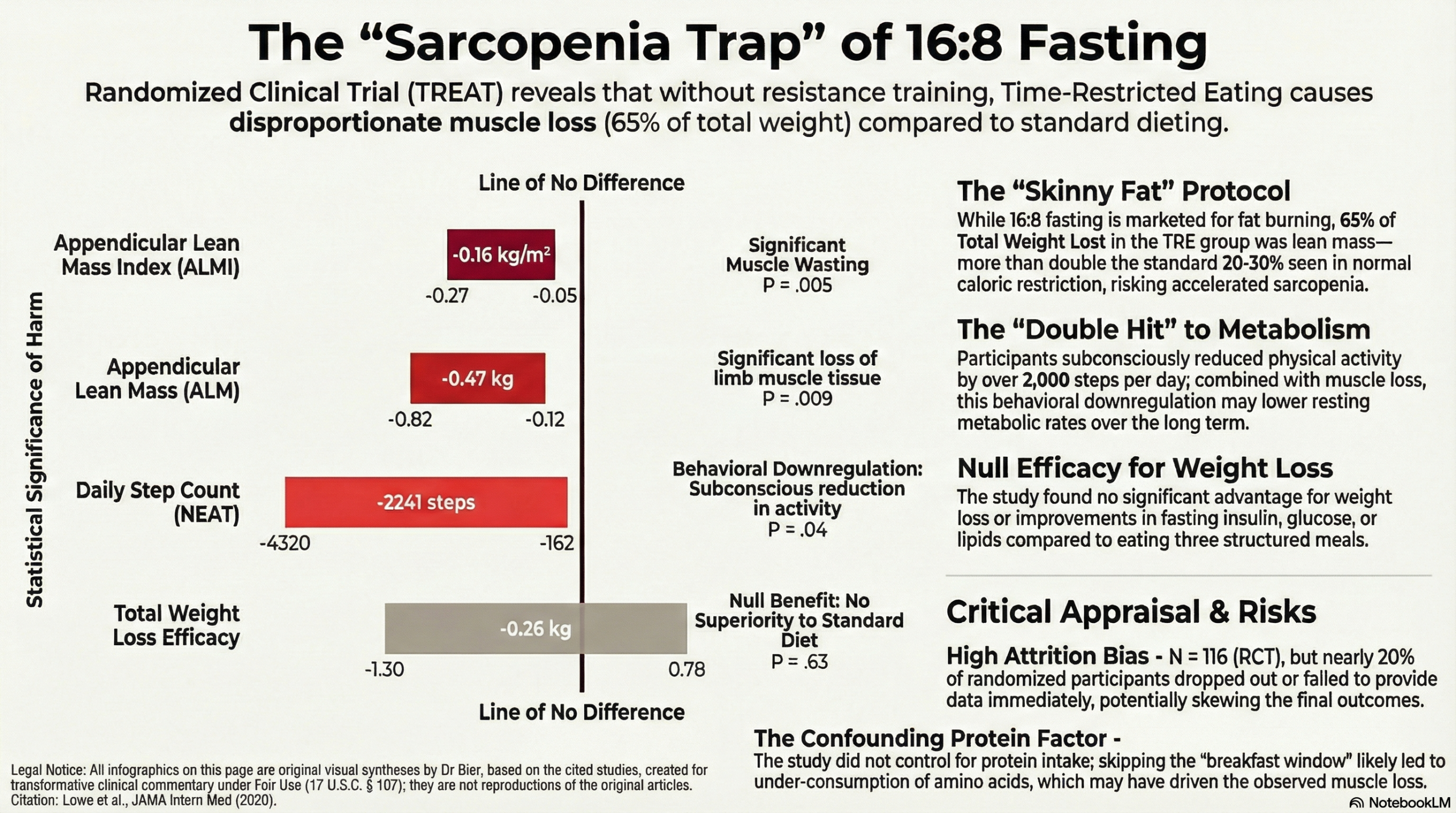
The specific safety signal identified in this cohort is critical for clinicians. While there was no significant difference in total weight loss between groups, the composition of that loss in the TRE group was alarming. In the sub-cohort assessed via DEXA scans, approximately 65% of the weight lost was lean mass—a figure that far exceeds the expected 20–30% ratio typically seen in standard caloric restriction. This indicates that without specific protein pacing or resistance training, a 16:8 protocol may inadvertently accelerate sarcopenia while chasing a lower number on the scale.
We must also look at the behavioral mechanism driving this muscle loss. The data showed a significant reduction in daily step counts for the TRE group, suggesting that the body may subconsciously downregulate non-exercise activity thermogenesis (NEAT) to conserve energy during the fasting window. This creates a “double hit” to metabolic health: the patient is moving less and simultaneously catabolizing the metabolically active tissue required to burn glucose. The physiological signal here is one of stress and conservation, not optimization.
As always the Precautionary Principle should be our guide, particularly for older adults or those with lower muscle reserves. A weight loss intervention that disproportionately sacrifices lean tissue is a pyrrhic victory. The burden of proof rests on the protocol to demonstrate that it can preserve muscle mass; until then, we must treat unsupported 16:8 fasting as a potential catabolic stressor.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
This randomized clinical trial challenges the utility of 16:8 Time-Restricted Eating (TRE) as a standalone weight loss intervention. The study found no significant difference in weight loss between patients assigned to TRE (eating 12:00 PM–8:00 PM) and those eating three structured meals per day (Control).
Crucially, regarding risks: The study identified a significant safety signal regarding body composition. In the cohort undergoing detailed body composition analysis, the TRE group experienced a disproportionate loss of lean muscle mass. Approximately 65% of the weight lost in the TRE group was lean mass—far exceeding the expected 20–30% typically seen with weight loss. This suggests that TRE, without specific instructions to maintain protein intake or perform resistance training, may carry a risk of sarcopenia (muscle loss).
Results in Context
Primary Outcome: Weight Loss
- Finding: There was no statistically significant difference in weight loss between the TRE group and the Consistent Meal Timing (CMT) control group after 12 weeks.
- Data: The TRE group lost an average of 0.94 kg, while the CMT group lost 0.68 kg. The between-group difference was -0.26 kg (95% CI, -1.30 to 0.78; P=.63), which is clinically negligible.
Key Secondary Outcome: Lean Mass (The Primary Risk Identified)
- Finding: In the in-person sub-cohort (n=50) assessed via DEXA scans, the TRE group lost a significant amount of appendicular lean mass (ALM) compared to the control group.
- Data:
- Total Weight Loss breakdown: The TRE group lost 1.70 kg total; of this, 1.10 kg was lean mass.
- Appendicular Lean Mass (ALM): There was a significant difference in ALM between groups (-0.47 kg difference; P=.009).
- Appendicular Lean Mass Index (ALMI): The difference between groups was also significant (P=.005).
- Context: The authors note that losing lean mass at a rate of 65% of total weight loss is highly unusual and “far exceeds the normal range”. This reduction in ALM can lead to weakness and disability, serving as a caution for populations at risk for muscle wasting.
Metabolic Outcomes
- Finding: There were no significant benefits of TRE over the control group for metabolic markers.
- Data: No significant differences were found in fasting glucose, fasting insulin, HOMA-IR, HbA1c, triglycerides, total cholesterol, HDL, or LDL levels.
Adherence and Activity
- Adherence: Self-reported adherence was high in both groups (TRE: 83.5%; CMT: 92.1%).
- Activity: There was a significant decrease in daily step counts in the TRE group compared to the control group (P=.04).
Assertive Critical Appraisal
Risk of Bias (RoB 2 Framework)
- Judgment: Some Concerns.
- Attrition/Missing Data: Of the 141 randomized participants, 25 (18%) never provided any data and were excluded from the primary analysis. While the authors used a linear mixed model to handle missing data for the remaining participants, the complete loss of nearly 20% of the randomized cohort immediately after assignment introduces a risk of attrition bias.
- Lack of Dietary Control: The study was designed as a “prescription” intervention rather than a feeding study. Participants did not receive calorie or macronutrient targets . While this increases “real-world” applicability, it is a major confounder for the adverse muscle loss finding. It is highly likely that the TRE group spontaneously reduced protein intake or skipped protein-rich meals (like breakfast), leading to the observed muscle wasting.
Appraisal of the Lean Mass Finding
- Hydration Confounding: DEXA scans can be influenced by hydration status. The authors acknowledge this limitation but argue that the magnitude of lean mass loss (1.10 kg) exceeded total body water loss, suggesting true muscle tissue loss occurred.
- Clinical Implication: This finding strongly suggests that clinicians prescribing TRE should likely pair it with resistance training and explicit protein intake guidelines to mitigate muscle loss.
Applicability
- The study is highly applicable to general clinical practice as it mimicked a standard advice-based intervention (“eat between 12 and 8”) without intensive dietetic support. The results suggest that simple advice to perform TRE without nutritional support is ineffective for weight loss and potentially harmful to muscle mass.
Research Objective
To determine the effect of 16:8-hour time-restricted eating (TRE) on weight loss and metabolic risk markers compared to a consistent meal timing (CMT) schedule.
Study Design
- Design: 12-week Randomized Clinical Trial (RCT).
Intervention:
- TRE Group: Ad libitum eating from 12:00 PM to 8:00 PM; fasting from 8:00 PM to 12:00 PM the next day.
- CMT (Control) Group: 3 structured meals per day.
- Blinding: Examiners collecting in-person data were blinded, but participants were aware of their assignment (unblinded) [implied by intervention type].
Setting and Participants
- Setting: United States (participants lived anywhere), with a subset of 50 participants near San Francisco undergoing in-person metabolic testing.
- Population: 116 analyzed adults (Men and Women) aged 18 to 64 years.
- Key Eligibility: BMI between 27 and 43.
Bibliographic Data
- Title: Effects of Time-Restricted Eating on Weight Loss and Other Metabolic Parameters in Women and Men With Overweight and Obesity: The TREAT Randomized Clinical Trial
- Authors: Lowe DA, Wu N, Rohdin-Bibby L, et al.
- Journal: JAMA Internal Medicine
- Year: 2020
- DOI: 10.1001/jamainternmed.2020.4153
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
