Comment:
For the last 30 years of practice, I have consistently recommended moving thyroid medication—including Desiccated Thyroid Extract (DTE)—to breakfast rather than fasting, often to the shock of conventional endocrinologists. This was based on original trials showing that coingestion with food reduced absorption, a factor I knew we could safely manage by monitoring the dose. This randomized trial provides the definitive clinical stability data for this approach, showing no significant difference in TSH, free T4, or total T3 levels when the dose is adjusted upward.
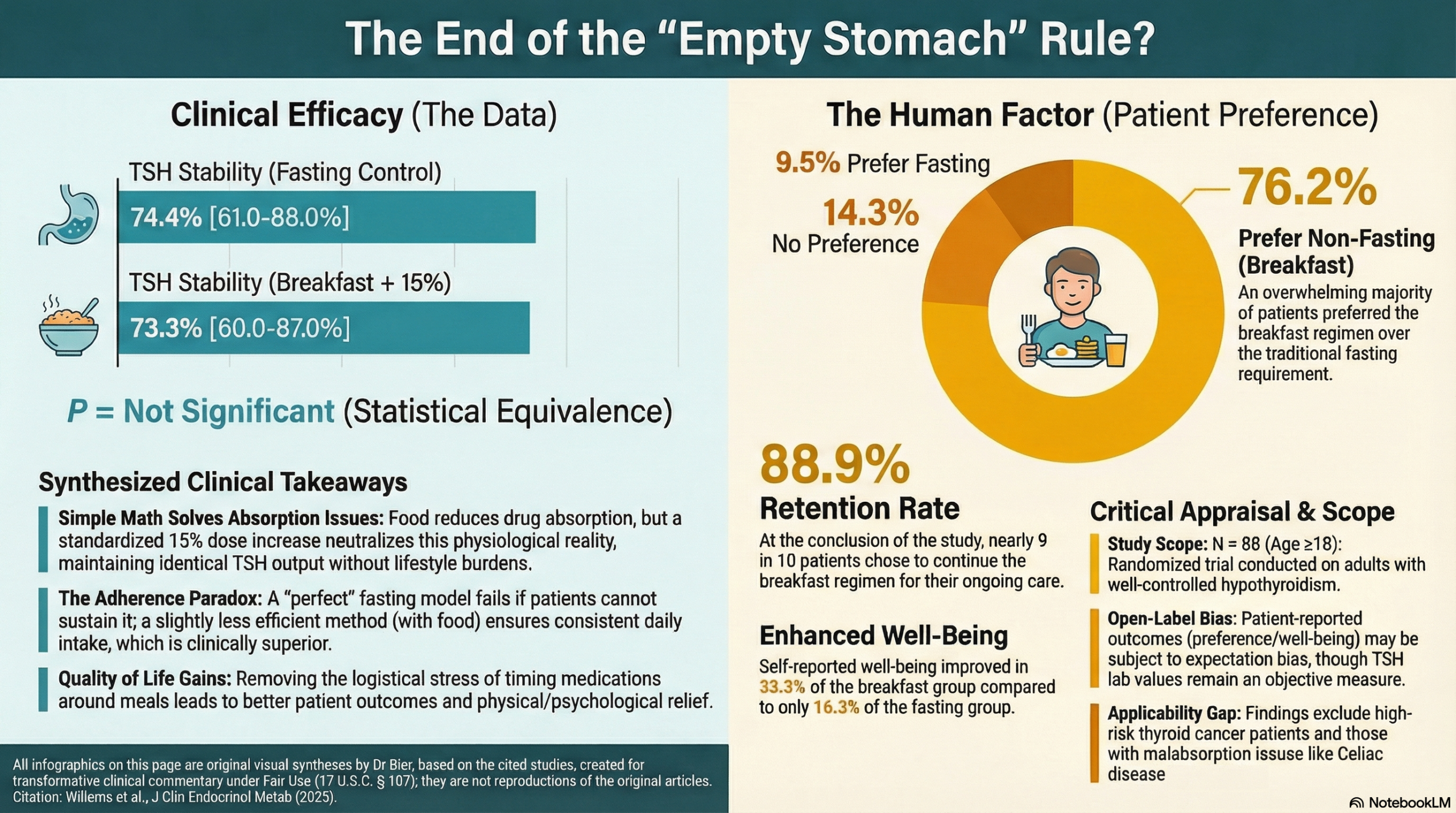
The hierarchy of harm here is unambiguous. While the conventional recommendation worries about a 15% absorption loss, the greater risk to systemic health is the nonadherence and “skipping” that occurs when patients find the fasting requirement burdensome. When 76.2% of patients prefer taking their medication with breakfast and 88.9% choose to continue the nonfasting regimen after the study ends, the clinical signal is clear – it is perfectly fine to take thyroid medication with food as long as it’s done consistently.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
This randomized trial provides strong proof-of-concept evidence that taking levothyroxine (LT4) with breakfast—when accompanied by a compensatory 15% dose increase—is as effective at maintaining TSH stability as the traditional fasting administration. Furthermore, this patient-centered approach significantly improved self-reported well-being and was overwhelmingly preferred by patients. For clinicians, this suggests that the rigid “fasting” requirement can be safely relaxed for stable patients, provided the dose is adjusted upward and monitored.
Results in Context
- Primary Outcome: TSH stability (defined as two consecutive TSH values within range and ≤1 mIU/L from baseline) was achieved by 74.4% of the fasting group and 73.3% of the breakfast group (P = not significant).
- Secondary & Specialized Outcomes:
- Patient-Reported Outcomes (PROs): Well-being improved in 33.3% of the breakfast group vs. 16.3% of the fasting group. Reasons for improvement included increased physical fitness, psychological relief from fasting burdens, and reduced stomach discomfort.
- Patient Preference: 76.2% of patients in the breakfast group preferred nonfasting intake, and 88.9% chose to continue this regimen after the study ended.
- Harms and Adverse Events: No adverse events were observed during the study period.
Assertive Critical Appraisal
- Risk of Bias (RoB 2 Framework): Some concerns. As an open-label trial, both patients and investigators were aware of the treatment allocation. While TSH is an objective laboratory marker (low risk for detection bias), the patient-reported outcomes (well-being and preference) are highly susceptible to performance and expectation bias.
- Reporting Quality Assessment (CONSORT): The study quality is high, including a clear participant flow diagram (Figure 1) and a detailed description of the randomization process.
- Appraisal of Patient-Reported Outcomes (CONSORT-PRO): The use of the validated ThyPRO-39 questionnaire in the crossover group strengthens the findings. However, the primary analysis for well-being was based on a non-validated assessment of “improved, stable, or declined,” which is less robust.
- Applicability: These findings are highly relevant to general clinical practice. The study used standard LT4 brands and a diverse hypothyroid population (primary, post-surgical, and post-ablative). However, results should not yet be extrapolated to pregnant patients, those with malabsorption disorders, or those with high-risk thyroid carcinoma, as these groups were excluded.
- The “15% Rule”: The 15% dose increase was an empirical choice to offset the known reduction in absorption caused by food. This provides a practical starting point for clinicians attempting this switch in practice.
Research Objective
To evaluate whether nonfasting LT4 intake—accompanied by a 15% dose increase—can maintain TSH stability compared to traditional fasting intake in adults with well-controlled hypothyroidism.
Study Design
An open-label, randomized controlled trial with a crossover component.
- Participants: 88 adults (80.7% female, median age 62).
- Follow-up: Every 6 weeks for up to 24 weeks.
Setting and Participants
- Setting: Zuyd Thyroid Center, Zuyderland Medical Center, The Netherlands.
- Eligibility: Adults on ≥1.0 mcg/kg LT4, eating breakfast ≥5 days/week, with stable baseline TSH.
- Exclusions: Pregnancy, malabsorption (e.g., Celiac), or high-risk thyroid cancer.
Bibliographic Data
- Title: Fasting vs Nonfasting, Dose-adjusted Levothyroxine Ingestion in Hypothyroidism: A Randomized Clinical Trial
- Authors: Jeresa I. A. Willems, Daan J. L. van Twist, et al.
- Journal: The Journal of Clinical Endocrinology & Metabolism
- Year: 2025
- DOI: https://doi.org/10.1210/clinem/dgaf686
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
