Comment:
This systematic review of 33 studies and over 2.5 million women reveals a critical tension between biochemical plausibility and actual clinical outcomes, at least for oral dosing. While observational data suggests early initiation reduces coronary heart disease, we should recognize that some of these findings are likely compromised by Healthy User Bias—where healthier women with better baseline metabolic profiles were the primary users, artificially inflating the therapy’s perceived benefits.
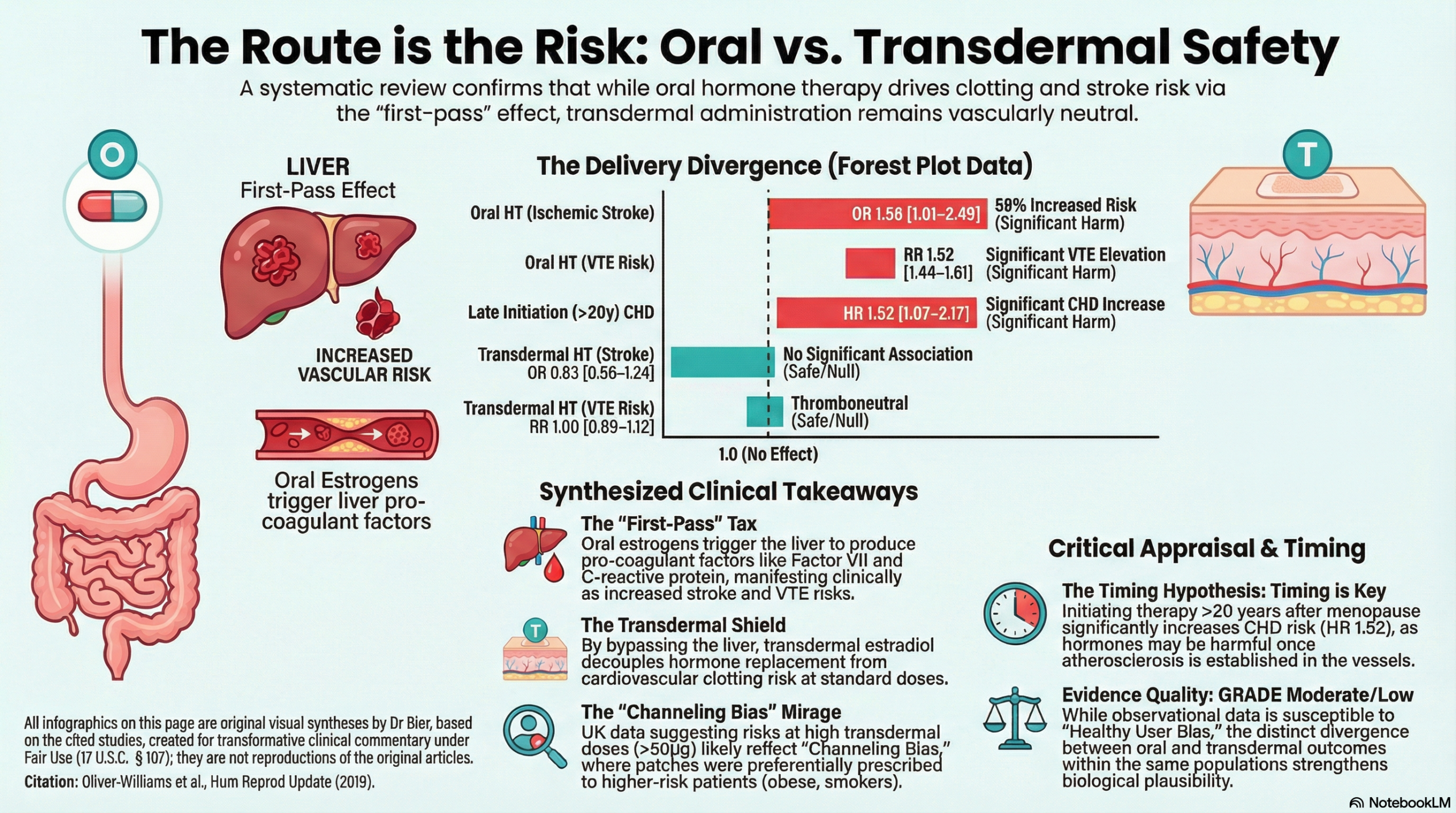
Regardless of timing, oral therapy remains a potent driver of cerebrovascular catastrophe. This route subjects the patient to a “first-pass metabolic tax” as the liver processes the estrogen, triggering systemic hypercoagulability through heightened thrombin generation and induced activated protein C resistance. This results in a 58% increased risk for ischemic stroke, escalating to a more than twofold risk at high doses (OR 2.41). Furthermore, the type of progestogen is a critical determinant of harm; specifically, norpregnane derivatives (such as nomegestrol acetate) are associated with a staggering 2.25-fold increased risk of stroke, a clear signal of iatrogenic hazard.
As opposed to oral dosing, the results with transdermal are more nuanced. While transdermal delivery is often marketed as a universally benign alternative, we should recognize that its perceived safety profile is not consistent across all datasets. The conflict lies in a critical discrepancy between the UK and French observational cohorts: while the French data (Canonico et al.) reported that stroke risk remained neutral even at higher doses, the UK data (Renoux et al.) identified a clear signal of harm, with an 1.89-fold increased stroke risk once transdermal doses exceeded 50 micrograms. This may be due to the opposite of a healthy user bias. In the UK, clinicians may have preferentially channeled higher-risk patients—those with elevated BMI or smoking histories—toward transdermal patches, whereas the French cohort likely benefited from a more pronounced “healthy user bias.” The registries often lack granular data on baseline metabolic variables and smoking status, so the authors attribute the difference to “diversity in scientific rigor and quality”. Which is correct, only the future will tell.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
This systematic review suggests that the cardiovascular risk profile of hormone therapy (HT) is highly dependent on the route of administration and the “timing” of initiation relative to menopause onset. Transdermal estradiol (typically <50micrograms/day) combined with micronized progesterone appears to be the safest regimen, particularly for women with elevated baseline thrombotic risk, as lower levels do not appear to increase the risk of venous thromboembolism (VTE) or stroke, although higher (>50microg/day) do although that is based on a small number of people from a single observation study. Conversely, oral HT is associated with a dose-dependent increase in stroke and VTE risk. Regarding timing, initiating HT within 10 years of menopause onset (or before age 60) may offer cardioprotective benefits and carries a lower absolute risk of adverse events compared to late initiation (>10 years post-menopause), where the risk of coronary heart disease (CHD) and stroke increases. The certainty of this evidence is generally Moderate (GRADE B) for most major outcomes, though it is heavily reliant on observational data.
Results
Summary of Results
- Route of Administration: Oral HT was associated with a 1.25- to 1.48-fold increased risk of stroke and a 1.52- to 4.2-fold increased risk of VTE. Transdermal HT showed no significant association with increased stroke or VTE risk at standard doses.
- Timing of Initiation: HT started within 10 years of menopause was associated with reduced CHD and mortality (RR 0.54 to 0.70 in some cohorts). Initiation >10 years post-menopause was associated with increased absolute risks of CHD, stroke, and VTE.
- Statistical Terms: The review utilizes Relative Risk (RR) and Odds Ratios (OR) to compare event rates; for instance, an OR of 1.58 for stroke with oral HT indicates a 58% increase in the odds of a stroke occurring compared to non-users.
| Dose Category | Oral HT Risk Profile | Transdermal HT Risk Profile |
|
Low Dose (≤ 0.3-0.625 mg CEE or equivalent) |
Elevated Risk Even at lower doses, oral therapy is associated with increased risk of Venous Thromboembolism (VTE) and Stroke compared to non-users. The “first-pass” hepatic metabolism activates coagulation factors regardless of the lower dose. |
Thromboneutral (Safe) Consistently shows no increased risk for VTE or Stroke. Avoiding the liver bypasses the activation of clotting factors (e.g., C-reactive protein, Factor VII), maintaining a neutral safety profile. |
|
Medium Dose (0.625 mg CEE / 50 µg Patch) |
Dose-Dependent Risk Increase Risk of VTE and Stroke rises linearly. Sweetland et al. found a 2-fold increased risk of VTE for oral users, with higher risks for those using Medroxyprogesterone Acetate (MPA). |
Thromboneutral (Safe) At the standard 50 µg dose, the consensus across major observational studies (French and UK cohorts) is that transdermal estradiol remains safe regarding VTE and stroke risk. |
|
High Dose (> 0.625 mg CEE / > 50 µg Patch) |
High Risk Strong, consistent signal for harm. Stroke risk can increase by ~1.5 to 2.4-fold depending on the specific study (Renoux, Grodstein). |
⚠️ The “Gray Zone” (Conflicting Data) UK Data: Renoux found a 1.89-fold increased stroke risk for doses >50 µg. French Data: Canonico found NO increased risk, even at high doses. Note: The “High Risk” signal relies on a small sample size of high-dose users in the UK database. |
Assertive Critical Appraisal
Certainty of Evidence (GRADE Framework)
The authors applied the GRADE approach, rating the evidence for most findings as Moderate (B) or Low (C).
- Downgrading Reasons: Evidence was primarily downgraded because it relies heavily on observational studies (which start as “Low” quality in GRADE) and exhibits some inconsistency in stroke outcomes across different HT formulations.
- High Quality Exception: The association between short HT duration and VTE risk was rated as High (A) certainty.
Heterogeneity
The review notes substantial diversity in HT formulations (estrogen-only vs. combined), doses, and study populations. While a specific I2 value for a single pooled meta-analysis is not provided (due to the qualitative nature of the synthesis for many outcomes), the authors explicitly state that “substantial diversity in scientific rigor and quality” made synthesis challenging. This indicates significant clinical and methodological heterogeneity, meaning results should be interpreted as general trends rather than precise universal estimates.
Publication Bias
The authors did not explicitly report a formal statistical assessment for publication bias (such as a Funnel plot or Egger’s test) in the provided text . Given that negative or “null” findings in HT research have historically been highly publicized, the risk may be lower than in other fields, but its absence remains a limitation in the transparency of the review process.
Risk of Bias in Included Studies
- Trials: 4 out of 6 included randomized controlled trials (RCTs) demonstrated medium or high risk of bias in at least one domain.
- Observational: Only 1 of 27 observational studies was considered at low risk of bias; 16 were rated as high risk. This underscores that the “cardioprotective” effects seen in observational data may be influenced by “healthy user bias”.
Reporting Quality Assessment (PRISMA)
The review adheres to PRISMA and MOOSE guidelines.
- Strengths: Includes a clear flow diagram (Fig. 1) showing the identification of 11,591 citations and the eventual inclusion of 33 unique studies.
- Weaknesses: The synthesis is largely qualitative for several endpoints due to heterogeneity, which, while appropriate, limits the ability to provide a single “bottom-line” pooled effect size for the clinician.
Research Objective
To systematically review the role of dose, route of administration, timing of initiation, and duration of HT on cardiovascular risk (clinical and subclinical) among postmenopausal women.
Study Design
Systematic Review.
- Search: Medline, Web of Science, and Cochrane Central (up to January 30, 2018).
- Selection: RCTs and prospective observational studies comparing HT to placebo or no treatment.
Setting and Participants
- Total Studies: 33 unique studies (6 trials, 27 prospective observational).
- Total Participants: 2,588,327 women.
- Population: Mostly North American and European women; ages 30–94.
| Study Group (Primary Author) | Participants (n) | Country |
Primary Trial/Cohort Name
|
|
Randomized Trials
|
|||
| Angerer et al. (2001) | 321 | Germany | — |
| Harman et al. (2014) | 727 | USA | KEEPS Trial |
| Hodis et al. (2016) | 643 | USA | ELITE Trial |
| Hulley et al. (1998) | 2,763 | USA | HERS Trial |
| Somunkiran et al. (2006) | 60 | Turkey | — |
| WHI (Prentice et al.) | 27,347 | USA |
Women’s Health Initiative
|
|
Observational Studies
|
|||
| Arana et al. (2006) | 9,429 | Spain | BIFAP Database |
| Bretler et al. (2012) | 3,322 | Denmark |
Danish National Registries
|
| Canonico et al. (2007) | 881 | France | ESTHER Study |
| Canonico et al. (2016) | 15,305 | France | E3N Cohort |
| Carrasquilla et al. (2015) | 846 | Sweden | SHEP Study |
| Carrasquilla et al. (2017) | 88,914 | Sweden |
Pooled Swedish Cohorts
|
| Cauley et al. (1997) | 9,704 | USA | SOF Study |
| Chilvers et al. (2003) | 2,592 | UK |
East Midlands Case-Control
|
| Corrao et al. (2007) | 76,875 | Italy |
Lombardia Healthcare
|
| de Vries et al. (2006) | 9,390 | UK | GPRD |
| Ferrara et al. (2003) | 24,420 | USA |
Kaiser Permanente
|
| Grodstein et al. (1999) | 9,236 | Sweden |
Uppsala County Cohort
|
| Grodstein et al. (2000-08) | 40,000-70,000 | USA |
Nurses’ Health Study
|
| Heckbert et al. (1997) | 334 | USA |
Diabetic Woman Sub-study
|
| Henderson et al. (1988) | 8,881 | USA |
Leisure World Cohort
|
| Hernandez Avila (1990) | 1,827 | USA | — |
| Høibraaten et al. (1999) | 528 | Norway | — |
| Hsia et al. (2006) | 10,739 | USA |
WHI Observational
|
| Jick et al. (1996) | 210 | UK | — |
| Le Gal et al. (2003) | 815 | France | EVA Study |
| Lemaitre et al. (2002) | 864 | USA | GHC Cohort |
| Lokkegaard et al. (2008) | 421,712 | Denmark |
Danish National Registries
|
| Lokkegaard et al. (2017) | 980,003 | Denmark |
Danish National Registries
|
| Paganini-Hill (1988) | 8,881 | USA |
Leisure World Cohort
|
| Pentti et al. (2006) | 13,032 | Finland | OSTPRE Study |
| Renoux et al. (2010a/b) | 70,715 | UK | GPRD |
| Rosenberg et al. (1993) | 1,716 | USA |
Massachusetts Case-Control
|
| Schneider et al. (2009) | 30,048 | UK | GPRD |
| Sidney et al. (1997) | 2,176 | USA | — |
| Stram et al. (2011) | 71,661 | USA |
California Teachers Study
|
| Tavani et al. (2005) | 1,114 | Italy | — |
| Tuomikoski (2015/16) | 550,118 | Finland |
National Registry
|
| Varas-Lorenzo (2000) | 635 | UK | — |
Bibliographic Data
- Title: The route of administration, timing, duration and dose of postmenopausal hormone therapy and cardiovascular outcomes in women: a systematic review
- Authors: Clare Oliver-Williams, Marija Glisic, Sara Shahzad, Elizabeth Brown, Cristina Pellegrino Baena, Mahmuda Chadni, Rajiv Chowdhury, Oscar H. Franco, and Taulant Muka
- Journal: Human Reproduction Update
- Year: 2019
- DOI: 10.1093/humupd/dmy039
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
