Comment:
This study suggests that the ‘dangers’ of high-dose iodine are likely overstated. For the vast majority of the population the healthy thyroid handles high iodine intake with resilience. The adverse events highlighted here likely represent a failure of supporting cofactors—specifically Selenium and Iron—rather than direct toxicity. These nutrients act as the ‘safety valves’ for iodine metabolism. When they are depleted, therapeutic intake becomes a stressor. Clinically, this implies our goal should be to optimize these co-factors to ensure patients have the metabolic machinery required to utilize it without negative consequences.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line:
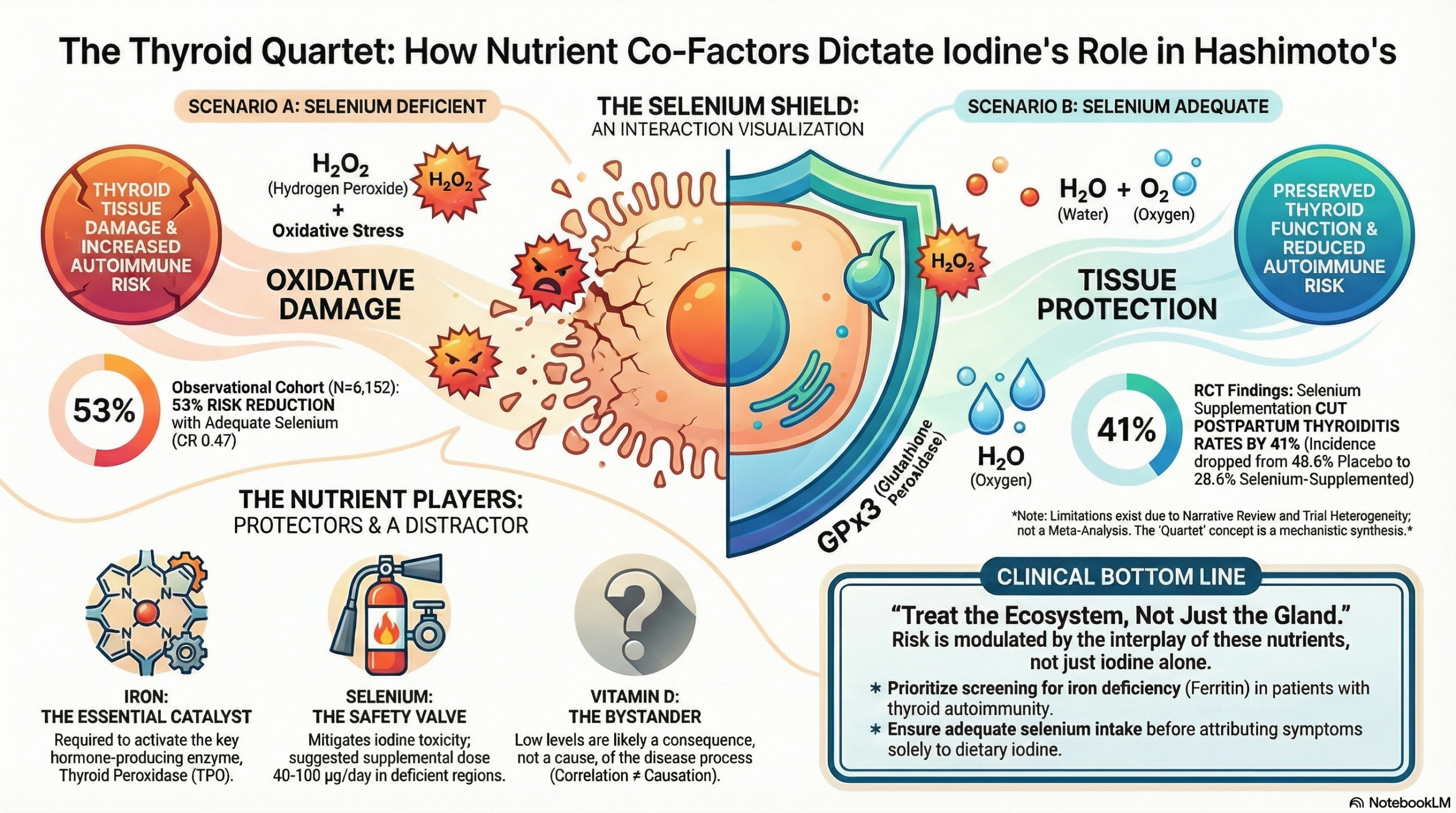
This narrative review argues that while iodine is essential for thyroid health, chronic excess intake is a well-established environmental trigger for Hashimoto’s Thyroiditis (HT), particularly in genetically susceptible subpopulations. The authors suggest that ensuring adequate selenium status is crucial, as selenoproteins protect the thyroid from oxidative damage caused by iodine processing. Additionally, clinicians should screen HT patients for iron deficiency (often due to co-morbidities like autoimmune gastritis) and correct vitamin D deficiency, though low vitamin D is likely a consequence rather than a primary cause of the disease.
Key Themes & Evidence Summary
1. Iodine: A Narrow Therapeutic Window
- Population Safety vs. Subpopulation Risk: Iodine intake determines the spectrum of thyroid disorders in a population. While severe deficiency causes goiter and hypothyroidism, the introduction of universal salt iodization or chronic excess intake is associated with a rise in thyroid autoimmunity.
- Mechanism of Risk: In genetically susceptible individuals, excess iodine increases the immunogenicity of thyroglobulin (Tg) and enhances auto-antigen presentation.
- Recommendation: To avoid increasing HT risk, iodine intake should remain within specific recommended ranges (e.g., WHO RNI of 150 μg/day for adults, 250 μg/day for pregnancy/lactation).
2. The Protective Role of Selenium
- Mechanism: Selenium is vital for glutathione peroxidases (e.g., GPx3), enzymes that protect thyrocytes by removing excessive hydrogen peroxide (H2O2) generated during thyroid hormone synthesis.
- Interplay with Iodine: Evidence from China suggests that in areas of adequate or high iodine, adequate selenium status is protective against autoimmune thyroiditis (OR=0.47).
- Clinical Efficacy: Meta-analyses indicate selenium supplementation can reduce TPO-antibody titers and improve well-being, though trial quality varies. It significantly reduced postpartum thyroiditis risk in one major RCT.
- Dosing: In regions of low intake (like Europe), a supplement of 50-100 μg/day is suggested, but caution is required as excess selenium is toxic.
3. Iron: Essential for Enzyme Function
- Physiology: Thyroid Peroxidase (TPO) is a heme-dependent enzyme; it requires iron to become active at the apical surface of thyrocytes.
- Clinical Association: Iron deficiency impairs thyroid hormone production. HT patients have a higher prevalence of iron deficiency, often driven by co-morbid autoimmune gastritis or celiac disease which impair absorption.
- Treatment: Treating iron deficiency can improve thyroid hormone concentrations, and combined T4/iron therapy is more effective than iron alone for improving iron status in hypothyroid patients.
4. Vitamin D: Correlation vs. Causation
- Observations: HT patients consistently show lower vitamin D status compared to controls, and levels inversely correlate with antibody titers.
- Causality Critique: The authors argue that low vitamin D is likely the result of the autoimmune process (e.g., Vitamin D Receptor dysfunction or sequestration in fat mass) rather than a cause. There is no strong RCT evidence that vitamin D supplementation prevents or treats HT.
Assertive Critical Appraisal
Evidence Level:
Narrative Review (Level 5 Evidence/Expert Opinion). This document summarizes existing literature but does not perform a new statistical meta-analysis. It is susceptible to selection bias, though the search strategy was defined.
Quality Assessment (SANRA Scale Audit):
- Justification & Aims: The review clearly defines its aim to elucidate the role of specific micronutrients in HT pathogenesis and treatment.
- Literature Search: The authors described a search of PubMed and the Cochrane Library using specific keywords, which increases transparency compared to many narrative reviews.
- Scientific Reasoning: The physiological arguments (e.g., the biochemical interaction between iodine load and selenoprotein protection) are logically presented and well-referenced. However, the authors appropriately note that for Vitamin D, correlation does not prove causation.
- Data Presentation: The review acknowledges the limitations of existing data, specifically noting that many selenium trials were flawed, underpowered, or lacked placebo controls.
- Bias Note: While the authors recommend selenium supplementation (50-100 μg/day) in low-intake regions, clinicians should note this is based largely on observational data and antibody-titer endpoints, rather than hard clinical outcomes (except for the postpartum thyroiditis data).
Research Objective
To elucidate the role of nutritional factors—specifically iodine, iron, selenium, and vitamin D—in the risk, pathogenesis, and treatment of Hashimoto’s thyroiditis.
Bibliographic Data
- Title: Multiple Nutritional Factors and the Risk of Hashimoto’s Thyroiditis
- Authors: Hu S, Rayman MP
- Journal: Thyroid
- Year: 2017
- DOI: 10.1089/thy.2016.0635
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
