The Wonk Debate – Audio Critique & Clinical Commentary:
Synthesized Clinical Bottom Line
updated 12-15-25
The relationship between iodine intake and thyroid pathology is complex, following a U-shaped curve where both deficiency and excess are detrimental. While correcting iodine deficiency is a public health priority, maintaining intake beyond “adequate” levels confers no additional clinical benefit and is consistently associated with a quantifiable, dose-dependent increase in subclinical thyroid dysfunction and autoimmune thyroiditis. Crucially, however, the long-term risk of overt, clinically significant disease does not significantly differ between “mildly deficient,” “more than adequate,” and “excessive” intakes. Much of the observed subclinical dysfunction is transient and reversible, reflecting the thyroid’s robust adaptive mechanisms. Nutritional cofactors—specifically Selenium and Iron—are essential for the thyroid’s enzymatic and antioxidant defense functions; deficiencies in these elements significantly lower the threshold for iodine-induced harm. Clinicians must prioritize correcting deficiency while concurrently ensuring sufficiency of these cofactors, especially in vulnerable groups
Thematic Evidence Analysis
- Safety and Risks: Subclinical Risk vs. Overt Stability
The evidence demonstrates a clear divergence, where biochemical abnormalities (which rise with intake) do not translate into a linear escalation of clinically overt disease (which remains stable).
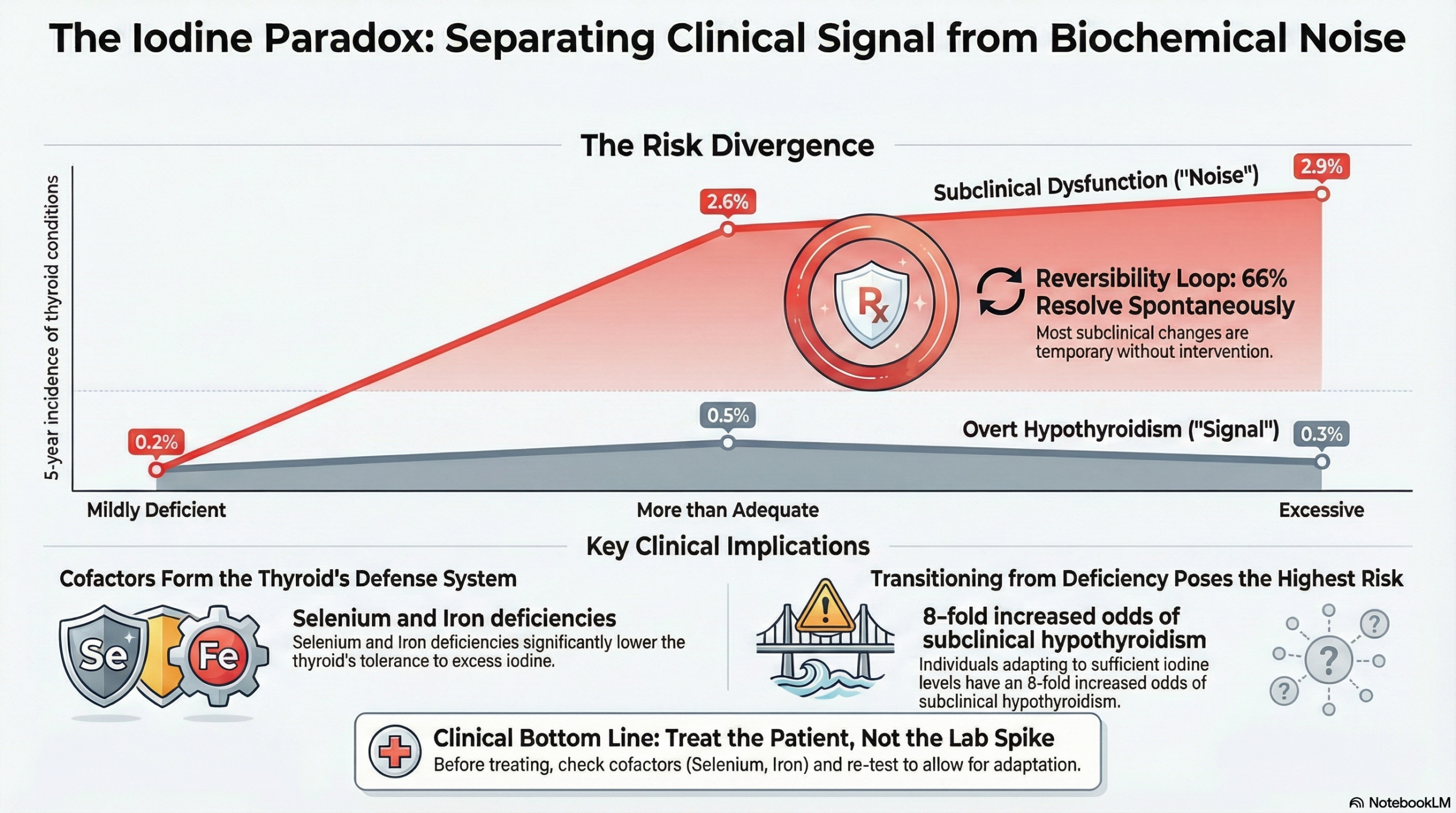
- The “More than Adequate” Risk Threshold: Risks for subclinical hypothyroidism and autoimmune thyroiditis emerge as soon as intake exceeds physiological requirements. High-quality observational data from a 5-year prospective cohort study in China demonstrated that risks emerge even at intake levels classified as “more than adequate” (Median Urinary Iodine Excretion [MUIE] 200–299 µg/L) versus “excessive” (MUIE ≥300 µg/L).
- Incidence of Subclinical Hypothyroidism: The cumulative incidence of subclinical hypothyroidism was significantly higher in populations with “more than adequate” intake (2.6%) and “excessive” intake (2.9%) compared to those with mildly deficient intake (0.2%) .
- Autoimmune Thyroiditis: The incidence of autoimmune thyroiditis was also elevated in populations with “more than adequate” (1.0%) and “excessive” (1.3%) intake compared to the deficient group (0.2%) .
- Plateau of Risk: No statistically significant difference in the incidence of subclinical hypothyroidism (P=0.68) or autoimmune thyroiditis (P=0.59) was observed between the “more than adequate” and “excessive” intake groups . This suggests a “threshold effect” where risk increases as intake moves from deficiency to sufficiency, but then plateaus rather than escalating linearly with greater excess .
- No Difference in Overt Disease: Crucially, the incidence of overt hypothyroidism remained low and statistically indistinguishable across all three groups over the 5-year period (Mildly Deficient: 0.2%; More than Adequate: 0.5%; Excessive: 0.3%). The investigators explicitly noted that “No significant difference in the cumulative incidence of overt hypothyroidism was observed among the three cohorts” .
- Clinical Significance, Adaptation, and Mechanisms of Injury
Many iodine-associated thyroid abnormalities appear to be transient biochemical phenomena, with the thyroid possessing robust adaptive mechanisms, though failure of these mechanisms is the root of pathology.
- Spontaneous Reversal: Evidence suggests a high rate of reversibility. In the same 5-year cohort, 66% of subjects diagnosed with subclinical hypothyroidism at baseline spontaneously reverted to a euthyroid (normal) state by the follow-up .
- The Wolff-Chaikoff Effect: The healthy thyroid adapts to acute iodine excess by temporarily inhibiting hormone synthesis (the Wolff-Chaikoff effect) to prevent thyrotoxicosis. In most individuals, the gland successfully “escapes” this inhibition by downregulating iodine transporters to resume normal function .
- Transient Epidemiology: Population-level data mirrors this individual adaptation. In Switzerland, the incidence of toxic nodular goiter and Graves’ disease rose immediately following salt iodization but subsequently decreased to levels lower than baseline over the following 16 years .
- Mechanisms of Iodine-Induced Injury: Excessive iodine-induced pathology is driven by three main mechanisms:
- Oxidative Stress: Excessive iodine processing generates redundant reactive oxygen species (ROS) and hydrogen peroxide (\text{H}_2\text{O}_2) . If not neutralized, this causes oxidative cell injury, triggering an immune response .
- Immunogenicity of Thyroglobulin (Tg): Excess iodine leads to the formation of highly iodinated thyroglobulin, which is more immunogenic and facilitates antigen presentation, promoting the development of thyroid autoantibodies .
- Wolff-Chaikoff Effect Failure: In susceptible individuals, the thyroid fails to “escape” this acute inhibitory effect, leading to persistent hypothyroidism .
- Nutritional Cofactors: The Protective Role of Selenium and Iron
The toxicity of excess iodine is heavily modulated by the presence of specific micronutrients that support the thyroid’s defense and enzymatic systems.
- Selenium (Se) – The Antioxidant Shield:
- Mechanism: Selenium is a required component of glutathione peroxidases (GPx), which are enzymes responsible for neutralizing the hydrogen peroxide (\text{H}_2\text{O}_2) produced during iodine processing .
- Clinical Impact: Selenium deficiency impairs this antioxidant defense, leading to thyroid tissue damage . Higher serum selenium is associated with significantly lower odds of autoimmune thyroiditis (OR 0.47) and hypothyroidism (OR 0.75) . Supplementation has been shown to reduce TPO antibody titers in pregnant women and decrease the incidence of postpartum thyroid dysfunction .
- Iron (Fe) – The Enzymatic Engine:
- Mechanism: Thyroid Peroxidase (TPO), the enzyme essential for thyroid hormone synthesis and organification, is heme-dependent and requires iron .
- Clinical Impact: Iron deficiency reduces TPO activity and is an independent risk factor for hypothyroxinemia . Treatment with Thyroxine (T4) plus iron has been shown to be more effective than iron alone in improving iron status in hypothyroid patients .
- Vitamin D: Low Vitamin D status is frequently observed in patients with Hashimoto’s thyroiditis, but the direction of causality (cause vs. consequence) remains unclear . Sufficiency is recommended for general immune regulation .
- Populations Warranting Caution
Specific groups lack the physiological reserve to adapt to “more than adequate” iodine loads and require careful monitoring.
- Populations Transitioning from Deficiency: Individuals moving from iodine-deficient to iodine-sufficient environments are at the highest risk for iodine-induced disorders. The shift from “mildly deficient” to “more than adequate” intake was identified as a significant risk factor for the development of subclinical hypothyroidism (Odds Ratio 8.0) . This transition can also trigger iodine-induced hyperthyroidism (Jod-Basedow effect) in those with autonomous nodules .
- Patients with Pre-existing Autoimmunity: Those with elevated thyroid antibodies at baseline are significantly more likely to progress to overt hypothyroidism when exposed to increased iodine loads .
- Neonates and Fetuses: The immature thyroid gland has a limited capacity to “escape” the Wolff-Chaikoff effect, making fetuses and neonates highly vulnerable to iodine-induced hypothyroidism .
- Lactating Women: High maternal iodine intake results in high breast milk iodine concentrations, posing a risk to the nursing infant.
- The Elderly: Older adults with long-standing nodular goiters are at specific risk for iodine-induced hyperthyroidism upon supplementation.
—–Overall Assessment of the Evidence
The evidence presented is robust, consisting of a large-scale prospective cohort study, comprehensive narrative reviews, and expert commentary . There is strong consensus that while correcting iodine deficiency is a public health priority, pushing intake levels beyond “adequate” (MUIE >200 µg/L) confers no additional benefit and measurably increases the risk of thyroid autoimmunity and subclinical hypothyroidism . The evidence strongly supports the protective role of selenium in mitigating oxidative stress within the thyroid .Gaps in the Evidence and Future Directions
- Selenium Dosage and Duration: While the benefit of selenium is established, the optimal dosing regimen and long-term safety of supplementation specifically for preventing iodine-induced damage require further randomized controlled trials .
- Vitamin D Causality: The direction of causality between Vitamin D deficiency and thyroid autoimmunity remains unresolved and requires prospective trial data .
- Long-term Outcomes of Subclinical Disease: The long-term clinical consequences of the mild, iodine-induced subclinical hypothyroidism observed in these populations need further longitudinal study to determine conversion rates to overt disease over decades .
List of Analyzed Papers
- Iodine Excess as an Environmental Risk Factor for Autoimmune Thyroid Disease (Luo et al., 2014)
- Excess iodine intake: sources, assessment, and effects on thyroid function (Farebrother et al., 2019)
- Effect of Iodine Intake on Thyroid Diseases in China (Teng et al., 2006)
- Multiple Nutritional Factors and the Risk of Hashimoto’s Thyroiditis (Hu & Rayman, 2017)
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
