Comment:
There is an assumption that specific routes of hormone therapy (MHT) offer a safe harbor from cardiovascular risk, yet this systematic review of over 5.5 million women identifies a profound disconnect between biochemical promise and systemic biological reality. While early initiation is frequently justified by observational data, we should recognize that these perceived benefits are heavily influenced by “healthy user bias,” where women starting therapy earlier often possess more favorable baseline metabolic profiles. This bias is further compounded by the study’s lack of data on smoking status and BMI—potent stroke risk factors that likely steered “healthier” patients toward transdermal options while oral routes were prescribed to those with higher baseline risks.
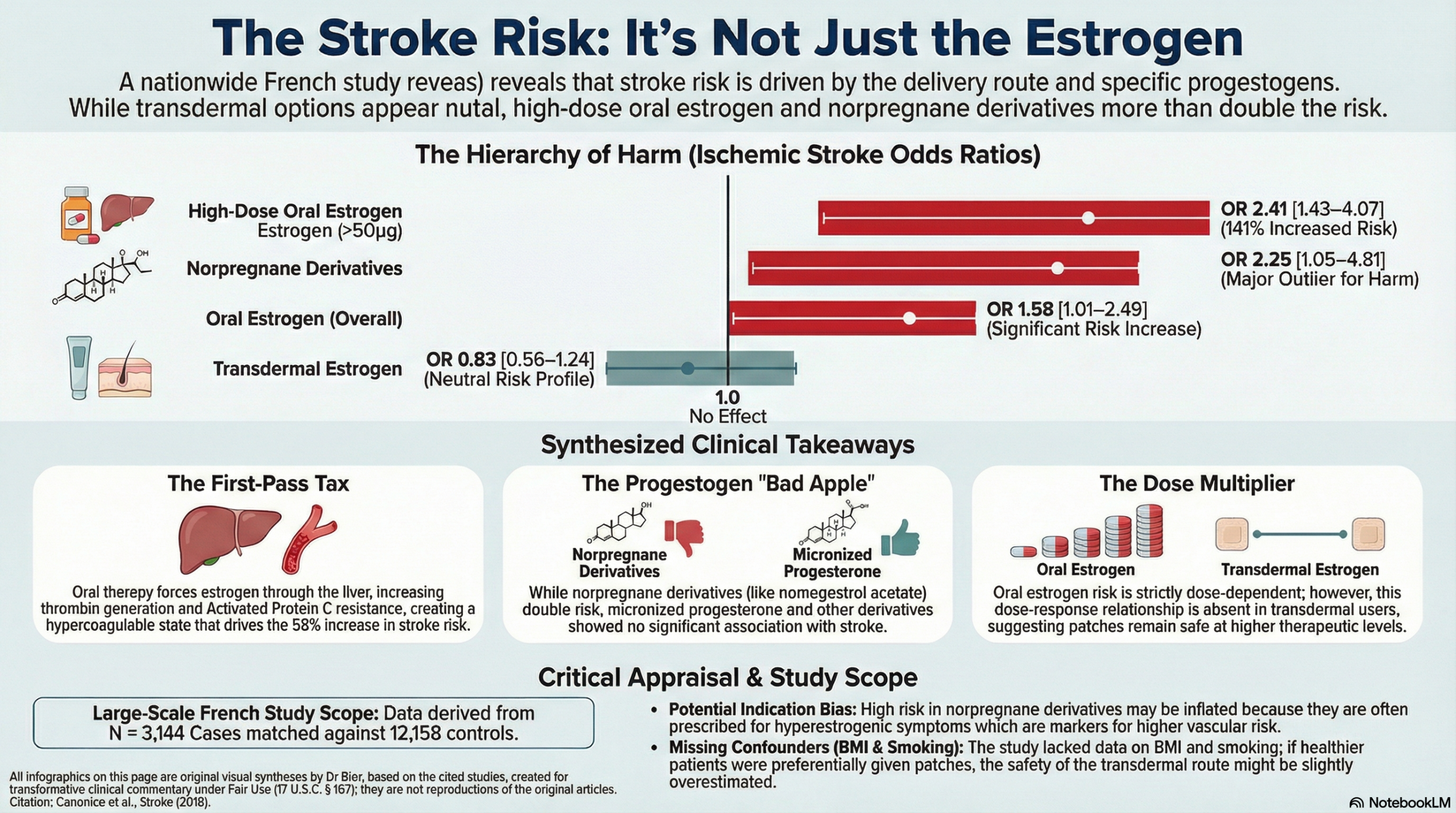
Regardless of timing, oral therapy remains a potent driver of cerebrovascular catastrophe. This route subjects the patient to a “first-pass metabolic tax” as the liver processes the estrogen, triggering systemic hypercoagulability through heightened thrombin generation and induced activated protein C resistance. This results in a 58% increased risk for ischemic stroke, escalating to a more than twofold risk at high doses (OR 2.41). Furthermore, the type of progestogen is a critical determinant of harm; specifically, norpregnane derivatives (such as nomegestrol acetate) are associated with a staggering 2.25-fold increased risk of stroke, a clear signal of iatrogenic hazard.
While this specific nested case-control data found transdermal delivery to be remarkably neutral—maintaining its safety profile even at the highest prescribed doses—this suggests that the harm is not inherent to estrogen, but an iatrogenic consequence of the oral delivery system and specific synthetic progestogens.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
This large nested case-control study suggests that the risk of ischemic stroke (IS) in postmenopausal hormone therapy (HT) depends heavily on the route of estrogen administration and the specific type of progestogen used. While oral estrogens were associated with a dose-dependent increase in stroke risk, transdermal estrogens appeared neutral. Furthermore, norpregnane derivatives (synthetic progestogens) were linked to a significantly higher risk, whereas micronized progesterone and other derivatives showed no such association. While these findings suggest transdermal estrogen combined with progesterone may be the safest profile, the observational design means we cannot rule out that these “safer” profiles were simply prescribed to “healthier” patients (healthy user bias).
Results in Context
- Main Results: Compared with nonusers, the adjusted odds ratio (OR) for IS was 1.58 (95% CI, 1.01–2.49) for oral estrogen users and 0.83 (95% CI, 0.56–1.24) for transdermal estrogen users.
- Definitions: An Odds Ratio (OR) of 1.58 means that the odds of having an ischemic stroke were 58% higher in the oral estrogen group than in the non-user group.
- Dose Response: For oral estrogens, the risk increased with dose: OR 1.39 for low dose, 1.84 for intermediate, and 2.41 for high dose (P for trend < 0.01). Transdermal estrogens showed no dose-response relationship.
- Progestogen Impact:
- Micronized Progesterone: OR 0.78 (95% CI, 0.49–1.26).
- Norpregnane derivatives: OR 2.25 (95% CI, 1.05–4.81).
- Participants: The study analyzed 3,144 ischemic stroke cases matched to 12,158 controls from a source population of over 5.5 million French women.
Assertive Critical Appraisal
Limitations & Bias (STROBE Framework)
- Confounding and Healthy User Bias: A major concern in HT research is that women prescribed transdermal therapy or micronized progesterone may have different baseline health profiles than those prescribed oral therapy. If “healthier” women (those with lower BMI or better exercise habits) were preferentially steered toward transdermal routes, the “protective” or neutral effect observed might be a reflection of their lifestyle rather than the drug itself.
- Unmeasured Confounders: The researchers used drug claims as proxies for hypertension, diabetes, and dyslipidemia. However, they lacked data on Smoking Status and Body Mass Index (BMI). Since both are potent risk factors for stroke and might influence which HT a doctor prescribes, their absence is a significant limitation.
- Indication Bias: The authors acknowledge that norpregnane derivatives are often used to treat hyperestrogenic symptoms like mastodynia, which may itself be linked to higher arterial risk, potentially inflating the risk attributed to the progestogen.
Reporting Quality Assessment (STROBE)
- Addressing Confounding: The authors did adjust for several major comorbidities (diabetes, hypertension, etc.) and found that the prevalence of these treated conditions was similar between oral and transdermal users. However, the omission of BMI and smoking remains a critical flaw in the reporting of potential confounders.
Reporting Quality Assessment (RECORD)
- Data Source Transparency: The study excels in describing the French National Health Insurance database. They clearly outlined the ICD-10 codes used for IS (Code 163) and the drug claim window (3 months) used to define “current use”.
Applicability
The findings are highly relevant to clinical practice, particularly the observation that transdermal estrogen does not appear to increase stroke risk even at higher doses. However, the specific progestogens used in France (e.g., nomegestrol acetate) differ from those commonly used in other regions (like MPA in the US), which may limit the generalizability of the progestogen-specific findings.
Research Objective
To investigate the association between ischemic stroke risk and postmenopausal hormone therapy, specifically comparing oral vs. transdermal estrogen and different pharmacological classes of progestogens.
Study Design
A nested case-control study using the French National Health Insurance database and hospital discharge data. Cases were matched to up to four controls based on age and zip code.
Setting and Participants
- Setting: France, nationwide health insurance data from 2009 to 2011.
- Participants: Women aged 51 to 62 years with no prior history of cardiovascular disease or contraindications to HT.
Bibliographic Data
- Title: Postmenopausal Hormone Therapy and Risk of Stroke: Impact of the Route of Estrogen Administration and Type of Progestogen
- Authors: Marianne Canonico, Laure Carcaillon, Geneviève Plu-Bureau, Emmanuel Oger, Archana Singh-Manoux, Pascale Tubert-Bitter, Alexis Elbaz, Pierre-Yves Scarabin
- Journal: Stroke
- Year: 2016
- DOI: 10.1161/STROKEAHA.116.013052
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
