Comment:
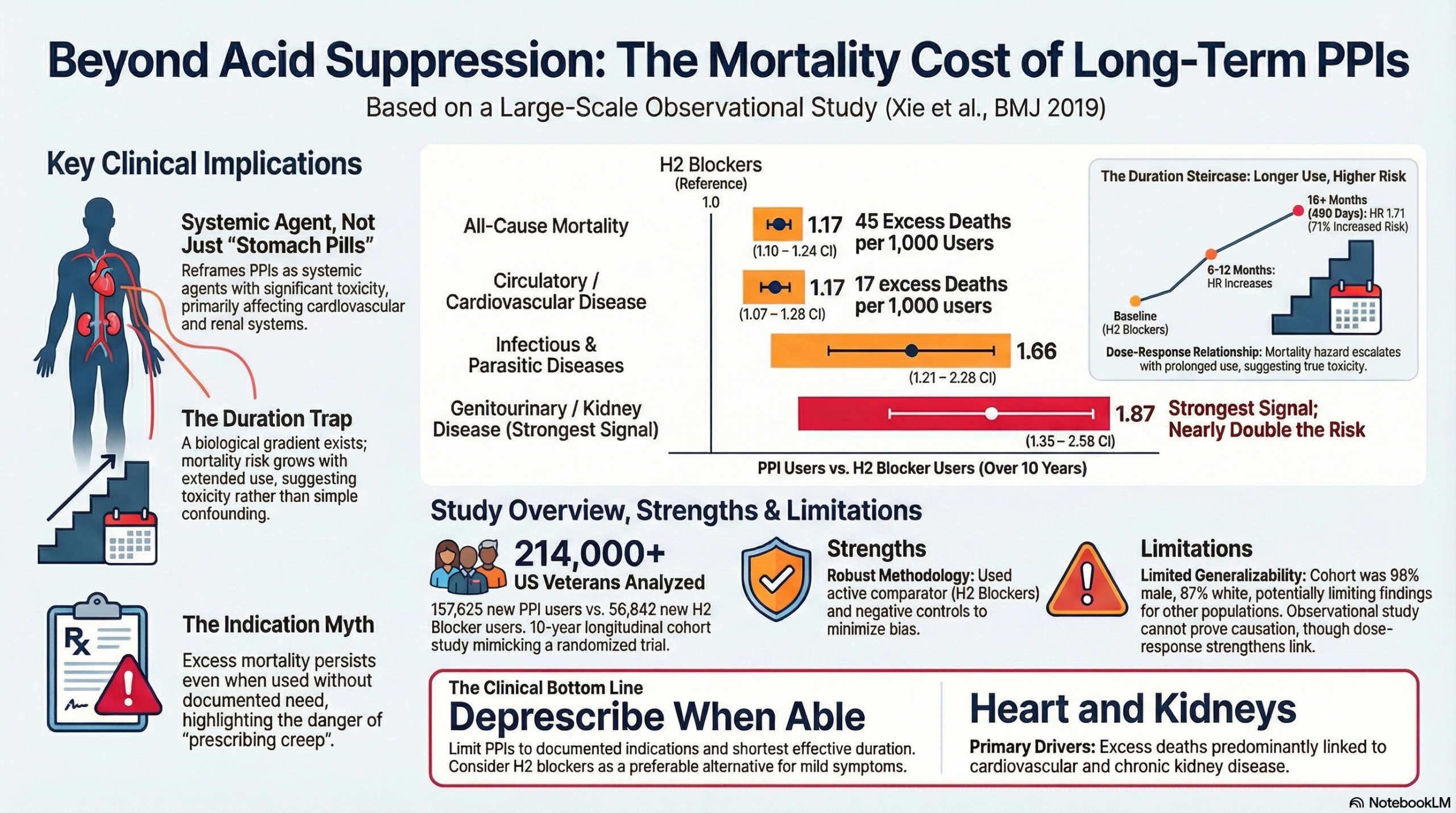
A hazard ratio of 1.17 for all-cause mortality is a statistic that shouldn’t be ignored, but unfortunately is. It translates to 45 excess deaths for every 1,000 patients treated. But the most concerning aspect of this data is the graded relationship: the longer the exposure, the higher the risk of death. This dose-response curve is a hallmark of true biological toxicity. It serves as a stark reminder that proton pump inhibitors are not merely ‘stomach pills’; they are systemic agents associated with significant cardiovascular and renal toxicity.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
This large observational cohort study demonstrates a statistically significant association between new proton pump inhibitor (PPI) use and excess all-cause mortality compared to H2 blockers, specifically driven by cardiovascular disease, chronic kidney disease, and upper gastrointestinal cancer. While the study employs rigorous methods to minimize confounding (including active comparators and high-dimensional propensity scores), it remains observational and cannot definitively prove causation. However, the graded relationship between duration of use and mortality risk strongly supports the clinical imperative to deprescribe PPIs when they are no longer medically indicated, particularly in older adults.
Results in Context
- Main Results:
- Excess Mortality: There were 45.20 excess deaths (95% CI 28.20 to 61.40) per 1,000 patients taking PPIs compared to H2 blockers over a 10-year follow-up.
- All-Cause Mortality: The Hazard Ratio (HR) for all-cause mortality was 1.17 (95% CI 1.10 to 1.24).
- Specific Causes: Significant excess burden was found for:
- Circulatory system diseases: Excess 17.47 deaths per 1,000 users; HR 1.17 (95% CI 1.07 to 1.28).
- Neoplasms: Excess 12.94 deaths per 1,000 users; HR 1.15 (95% CI 1.01 to 1.31).
- Genitourinary system diseases (including CKD): Excess 6.25 deaths per 1,000 users; HR 1.87 (95% CI 1.41 to 2.48).
- Infectious/Parasitic diseases: Excess 4.20 deaths per 1,000 users; HR 1.66 (95% CI 1.21 to 2.28).
- Duration Effect: The study found a graded relation; the risk of death increased with the cumulative duration of PPI exposure.
- Definitions:
- Hazard Ratio (HR): An HR of 1.17 indicates that at any given point during the study, the rate of death in the PPI group was 17% higher than in the H2 blocker group.
- Excess Burden: This refers to the calculated number of additional deaths attributable to the medication per 1,000 users, providing an absolute measure of risk rather than just a relative one.
- Participants:
- The study included 157,625 new users of PPIs and 56,842 new users of H2 blockers.
- A total of 214,467 patients were included in the final cohort.
Assertive Critical Appraisal
- Limitations & Bias (STROBE Framework):
- Demographics and Generalizability: The cohort consisted of US veterans who were predominantly male (95.93%), older (mean age 65.10), and white (87.43%). This significantly limits the applicability of the findings to women and younger populations.
- Residual Confounding: Despite using advanced statistical techniques, unmeasured confounding remains a risk inherent to observational designs.
- Exposure Measurement: Exposure was defined by prescription records (days supply), which does not guarantee the patient actually ingested the medication or account for over-the-counter PPI use.
- Reporting Quality Assessment (STROBE):
- The authors rigorously addressed confounding—the primary threat to observational studies. They utilized an active comparator (H2 blockers) to minimize indication bias. Furthermore, they employed High Dimensional Propensity Scores (HDPS) to balance covariates 1and instrumental variable analysis (physician prescribing preference) to account for unmeasured confounders. This represents a high standard of methodological reporting.
- The use of negative controls (transportation-related death and peptic ulcer disease death) where no association was found lends further validity to their results.
- Reporting Quality Assessment (RECORD) for RWE Studies:
- The study adequately describes data sources, utilizing the Department of Veterans Affairs corporate data warehouse and the National Death Index.
- Codes for outcomes (ICD-10) and exposures are clearly defined, adhering to RECORD principles.
- Applicability:
- While the biological mechanisms proposed (e.g., endothelial senescence, oxidative stress) likely apply generally, the specific magnitude of risk may differ in a general, non-veteran population with different baseline comorbidities and demographics.
Research Objective
To estimate all-cause mortality and cause-specific mortality among patients taking proton pump inhibitors (PPIs) compared to those taking H2 blockers.
Study Design
- Design: Longitudinal observational cohort study.
- Methodology: New user design with an active comparator (H2 blockers).
- Analysis: The study aimed to emulate a target randomized controlled trial using intention-to-treat principles, inverse probability of treatment weighting (IPTW), and instrumental variable analyses.
Setting and Participants
- Setting: US Department of Veterans Affairs healthcare system.
- Dates: New users identified between July 1, 2002, and June 30, 2004, followed for up to 10 years.
- Eligibility: Patients with no record of acid suppression drug prescription in the preceding 180 days (new users).
Bibliographic Data
- Title: Estimates of all cause mortality and cause specific mortality associated with proton pump inhibitors among US veterans: cohort study
- Authors: Yan Xie, Benjamin Bowe, Yan Yan, Hong Xian, Tingting Li, Ziyad Al-Aly
- Journal: BMJ
- Year: 2019
- DOI: 10.1136/bmj.l1580
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
